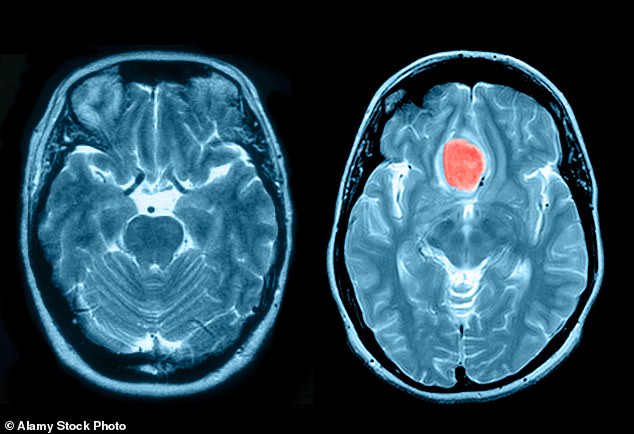
Women using a popular contraceptive injection face a heightened risk of developing potentially fatal brain tumors, according to a new study. This marks the third significant investigation in a little over a year that suggests the contraceptive, which increases the likelihood of meningioma— the most prevalent form of brain tumor— particularly in those who’ve been on the medication for 12 months or longer.
In England, approximately 10,000 prescriptions are written each month for medroxyprogesterone acetate, known by its brand name, Depo Provera. Following these findings, numerous women in the UK are reportedly exploring legal actions against the manufacturer, Pfizer, for allegedly not sufficiently warning them about these risks.
In the United States, a class action has already been initiated against Pfizer and other generic manufacturers. Over 500 women assert that these companies were aware of the associated risks but failed to provide adequate warnings or promote alternative contraceptives.
This injectable hormone, administered every three months, prevents ovulation. It was first approved as a contraceptive by the NHS over 40 years ago. Yet, a study published in 2024 in the British Medical Journal, which analyzed data from over 18,000 women, found that those who remained on the injection for at least a year were up to five times more likely to develop a meningioma.
Another investigation, conducted by the University of Alabama in September of last year, yielded similar conclusions. While meningiomas are typically non-cancerous, they can lead to blurred vision, headaches, hearing problems, a diminished sense of smell, and swallowing issues. Though they grow slowly, large tumors can press against the brain and nerves, posing serious risks.
In the UK, between 2,000 and 3,000 meningioma cases are diagnosed annually, with a higher prevalence in women. The latest research from the University of British Columbia in Canada compared the rates of meningioma in 72,181 women on Depo Provera with over 247,000 women utilizing oral contraceptives. The findings, published in the journal Expert Opinion on Drug Safety, indicated that long-term users of the injection had a risk more than three times greater than those who opted for pills.
Researcher Dr. Mahyar Etminan noted that, intriguingly, the risk doesn’t seem to increase with longer usage of the injection. He remarked, “I believe the evidence is now strong, with three substantial studies presenting similar results.” For women who are concerned about this potential risk, he suggested considering other contraceptive options.
The exact reason for the injection’s link to tumor growth remains unclear. One theory posits that the synthetic hormone, progestogen, may interact with meningioma cells, fostering their growth. Some studies indicate that certain formulations of the oral contraceptive pill containing progestogen might also elevate the risk for a small number of women, particularly if taken for over five years.
Last October, the UK’s Medicines and Healthcare Products Regulatory Agency urged Pfizer to warn users in patient information leaflets about this increased risk. Additionally, Pfizer communicated with NHS doctors, advising them to discontinue Depo Provera for any women diagnosed with meningioma.
Virginia Buchanan, a partner at the law firm Levin Papantonio—which is handling the case in the U.S.—stated, “This new study reinforces the need for legal action against Pfizer, in hopes of achieving justice for the women who were never informed of the increased risk of developing brain tumors while using Depo Provera and who have subsequently developed meningiomas.”
Pfizer has declined to make a comment.