Cell lines and animals
The HaCat, mouse hepatoma (AML-12), mouse embryonic fibroblast (3T3-L1), and mouse skeletal muscle (MSMC) cell lines were sourced from the Cell Bank of the Chinese Academy of Sciences. To create cells that express green fluorescent protein (HaCatGFP), lentivirus transfection of GFP plasmids was performed on HaCat cells as per the manufacturer’s instructions. All cell lines were maintained in a DMEM medium supplemented with 10% FBS and 1% penicillin–streptomycin, kept at 37°C in a 5% CO2 environment.
Male C57BL/6J mice (6–8 weeks old, around 25 g) and female Sprague–Dawley (SD) rats (8–12 weeks old, about 200 g) were obtained from Shanghai SLAC Laboratory Animal. These mice and rats were housed in the Laboratory Animal Center at Zhejiang University in specific-pathogen-free conditions. Male Guangxi Bama minipigs (6 months old, weighing between 35–40 kg) were bought from Shanghai Jiagan Laboratory Animal and were also kept at Zhejiang University under similar conditions. The animals were fed standard diets and maintained on a 12-hour light/dark cycle, with unrestricted access to water unless noted otherwise. The environment was controlled, maintaining temperatures between 20–26°C and relative humidity at 50–70%. All animal experiments were conducted according to the protocols approved by the Institutional Animal Care and Use Committee of Zhejiang University.
Experimental materials
Unless noted otherwise, all materials were purchased from Sinopharm Chemical Reagent. Dichloromethane (CH2Cl2) and tetrahydrofuran (THF) were purified over calcium hydride or treated with a 4 Å molecular sieve. Other chemicals like Trifluoroacetic acid, 2,2′-azobis(2-methylpropionitrile), and various other reagents were acquired from Energy Chemical. Fluorescein isothiocyanate and sulfo-cyanine dyes were sourced from Lumiprobe, and human recombinant insulin was purchased from Solarbio Science & Technology. Additional items included gold nanoparticles and various other compounds from different suppliers.
Synthesis of N-[2-(N-tert-butoxycarbonylamino)]ethyl-4-(dodecyltrithio-carbonate)-4-cyanopenteramide (N-Boc-CTA)
In a dry solution of CH2Cl2, CTA, EDC·HCl, and HOBT were mixed and stirred for 4 hours. Afterward, N-Boc-ethylenediamine and DIPEA were added, and the reaction proceeded overnight at room temperature. The resulting mixture was washed, dried, and subjected to column chromatography, yielding a yellow solid.
Synthesis of Boc-amino-terminated poly[2-(N,N-dimethylamino)ethyl methacrylate] (N-Boc-PDMA)
DMA, N-Boc-CTA, and AIBN were combined in THF within a Schlenk flask and purged with dry nitrogen. The polymerization proceeded at 65°C for 12 hours. Post-reaction, the solution was concentrated, precipitated in cold n-hexane, and subsequently dried.
Synthesis of Boc-amino-terminated poly[2-(N-oxide-N,N-dimethylamino)ethyl methacrylate) (N-Boc-OP)
N-Boc-PDMA was treated with hydrogen peroxide, stirred for 4 hours, and then dialyzed to remove any unreacted reagents. The final product was obtained after lyophilization.
Synthesis of OP-NH2
OP-NH2 was synthesized by dissolving N-Boc-OP in dichloromethane and trifluoroacetic acid. The solution underwent a reaction followed by a series of purification steps to yield the final compound.
Synthesis of OP-DBCO
OP-NH2 was mixed with DBCO-NHS under appropriate conditions, stirred for 4 hours, and purified by dialysis to obtain the product.
Synthesis of OP–I
Insulin was modified with an azide group and subsequently combined with OP-DBCO. The resultant mixture underwent purification, resulting in OP–I.
Labelling OP, OP–I, PEG–I and insulin with FITC, Cy3 or Cy5
Solutions of dyes were added to combinations of OP-NH2, PEG-NH2, insulin, and their conjugates, with reactions carried out overnight in darkness. Following the reactions, the labeled products were purified and stored appropriately for further use.
RP-HPLC analysis
RP-HPLC was conducted using specified equipment and conditions to evaluate the samples processed, with specific methodologies related to mobile phase compositions and flow rates standardly applied.
Stability of OP–ICy5
The stability of OP–ICy5 was assessed by incubating it in a culture medium and sampling at various time points to analyze its retention time and confirm structural integrity using HPLC methods.
Prep-HPLC for purification
The purification of compounds was carried out with defined methodologies, and retention times of the products were monitored as needed.
Gel-permeation chromatography
The method involved specific equipment and conditions focusing on the mobile phase compositions to analyze the samples effectively.
Circular dichroism spectroscopy
The far UV circular dichroism spectra were recorded with specific conditions regarding the path length and wavelengths utilized during the analysis.
MALDI-TOF MS analysis
MALDI-TOF MS was executed on specialized instruments, relying on specific matrices for the analysis, particularly focusing on the reduced forms of certain compounds for effective measurement.
Zeta potential measurements
Potentials were measured by preparing solutions at varied pH levels and analyzing them with designated equipment.
Synthesis of OP–AuNPs
OP-NH2 was reacted with gold nanoparticles in defined conditions to produce OP–AuNPs, which were then purified appropriately.
In vivo skin permeation of OPCy5, OP–IFITC, OP–ICy5 and their mixtures
A study was set up involving the application of solutions to mouse skin, with subsequent analyses of absorption and distribution conducted using various imaging techniques.
Biological half-life of OP–I
Half-lives were calculated following administration of solutions to mice with careful monitoring of blood samples collected over time.
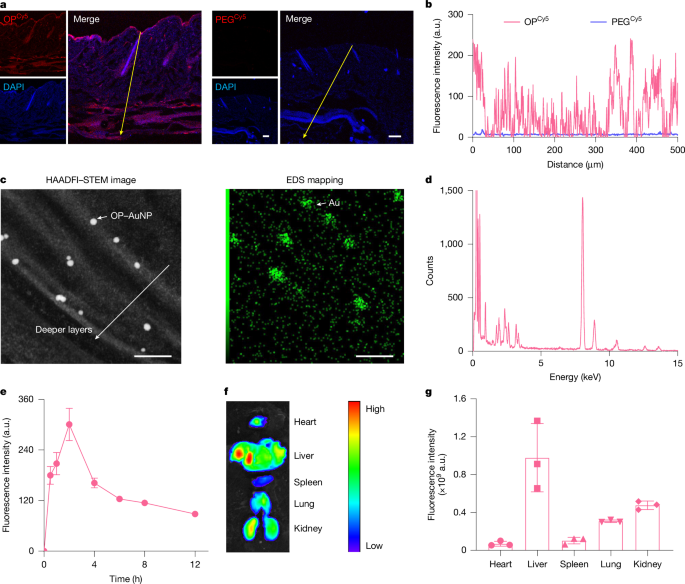
HAADFI–STEM-EDS analysis of OP–AuNP-treated skin cryosections
Mice underwent treatment before the skin was analyzed histologically to provide insights into the interactions of materials embedded within.
Cellular uptake
Cells were exposed to formulations at specific concentrations, and subsequent staining allowed for examination and visualization using confocal imaging techniques.
SPR analysis of binding affinity
Binding studies were performed on a designated instrument, allowing for evaluations of various concentrations to determine affinities.
Western blotting for insulin signalling analysis
Mice were injected with varied treatment solutions, with muscle tissues later collected for quantification of protein responses using established methodologies.
OP–I permeation in 3D skin equivalent EpiKutis
An in vitro skin model was constructed for the assessment of permeation, with populations of cells layered effectively for thorough investigation.
Skin penetration analysis by intravital two-photon microscopy
Mice treated topically underwent imaging facilitated by two-photon microscopy, further clarifying the distribution of treatments over a timeline.
Subcutaneous lymphatic vessel co-localization
Skin samples were examined following treatment applications, with lymphatic vessels highlighted for detailed imaging of treatment distribution.
Skin retention of OP–ICy5
Retention levels were indexed following specific treatment durations, with imaging performed to analyze fluorescence intensities remaining in skin samples post treatment.
In vivo skin permeation of OP–ICy5 in minipigs
Minipigs underwent treatments mirroring the mouse studies, with subsequent histological evaluations contributing to insights on permeation and retention.
OP–ICy5 biodistribution in mice
Following topical application, biodistribution analysis was conducted in various organs over time to gauge effective dissemination of the treatment.
In vivo studies using STZ-induced diabetic mice
Type 1 diabetic mouse models were established via specific interventions, allowing for detailed studies on the impact of treatments over blood glucose levels.
In vivo studies using STZ-induced diabetic minipigs
Minipigs were similarly treated to observe the effectiveness and duration of drug action, allowing critical insights into treatment efficiency for diabetes.
SC sample collection and observation
Samples from skin were collected and evaluated in accordance with previous methodologies, facilitating observations regarding the effectiveness of treatment penetration.
FTIR spectrometry
Studies using FTIR were executed with a focus on specific compounds, allowing for insights into the behavior of various combinations when interacting with skin samples.
MD simulations
MD simulations explored how insulin and its derivatives interacted within defined settings, providing computational support for experimental findings. Various factors and conditions affecting these interactions were tested.
PMF analyses
Potential mean force evaluations were carried out, utilizing advanced computational setups to scrutinize binding and diffusion characteristics of studied compounds within lipid membranes.
VE penetration study using 3D-cultured multilayer HaCat spheroids
Multilayer spheroids were created for testing, allowing for comparative analyses of different treatments applied under tightly controlled conditions.
Localization of OP–ICy5 at HaCat cell membranes
Localization studies employed various fluorescent tags to visualize the interactions at the cell membrane level, emphasizing treatment uptake over time.
Observation of contact-dependent direct transfer of OP–I among HaCat cells
Research on cellular transfer dynamics was performed through defined setups, focusing on how treatments could move between adjacent cells.
Non-contact inhibition of intercellular transfer of OP–ICy5
Experiments designed to explore how treatments affect one another’s transferability without direct contact were carried out to provide insights into cellular mechanics.
Intercellular transfer between cells on separate coverslips
Cell co-culture experiments provided a comparative platform for analyzing treatment transfer dynamics under closely controlled conditions.
TIRFM imaging
Detailed imaging utilizing TIRF methodologies provided insights into cellular handling of materials over time.
Statistical analyses
Statistical methods were applied for ensuring data integrity and significance in experimental findings.
Reporting summary
Further information on the study methodology is accessible in the Reporting Summary linked to the article.