FDA Expands Approval for Controversial Female Libido Drug
WASHINGTON — U.S. health officials have broadened the approval of a much-discussed medication aimed at enhancing female libido, now allowing its use for women over 65. This change, announced by the Food and Drug Administration, means that older women who have gone through menopause can now take the once-a-day pill.
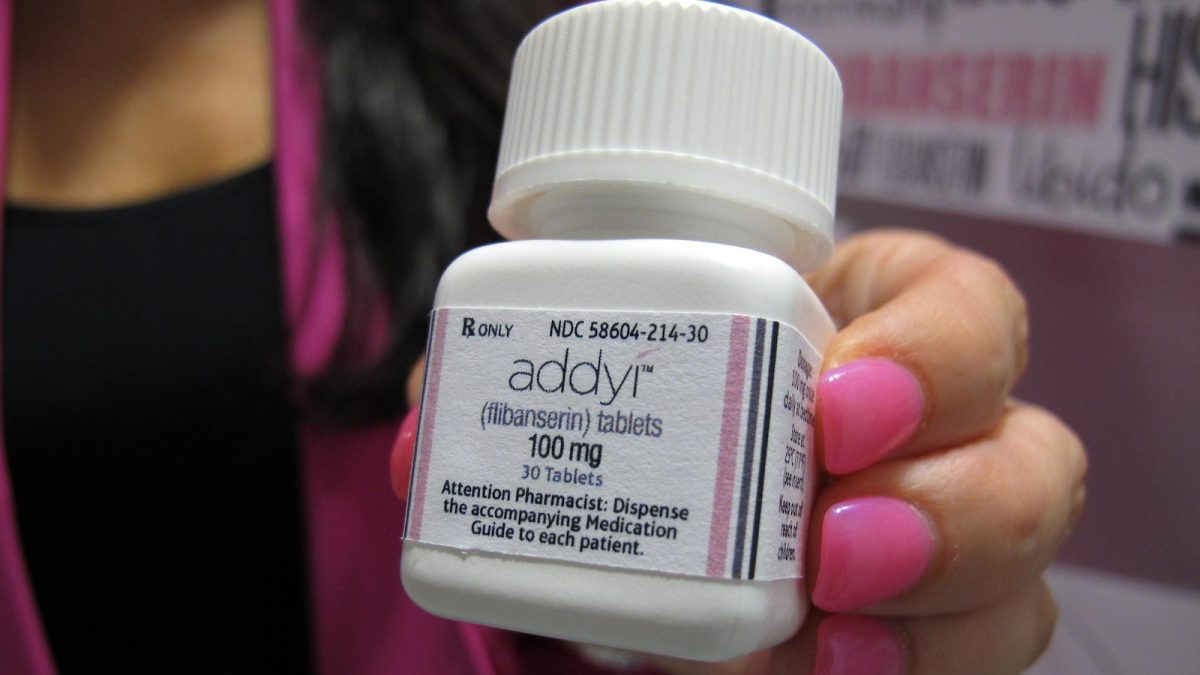
The medication, Addyi, was first approved a decade ago for premenopausal women who experience emotional distress due to low sex drive. Marketed by Sprout Pharmaceuticals, Addyi was initially viewed as a potential blockbuster that could address a significant gap in women’s health. However, it has come under scrutiny for side effects like dizziness and nausea, and it carries a serious warning about the risks of drinking alcohol while on the drug, including dangerously low blood pressure and fainting.
Sales of Addyi, which targets brain chemicals influencing mood and appetite, have not reached expected levels. In 2019, the FDA approved another drug that provides a different approach to treating low female libido — an on-demand injection acting on different neurological chemicals.
Sprout CEO Cindy Eckert expressed that the latest FDA decision “reflects a decade of persistent work with the FDA to fundamentally change how women’s sexual health is understood and prioritized.” The company, headquartered in Raleigh, North Carolina, shared the news in a press release on Monday.
Hypoactive sexual desire disorder, the medical term for low sexual appetite, has been acknowledged since the 1990s and is believed to affect many American women. Following the success of Viagra for men, pharmaceutical companies began investing heavily in researching potential treatments for sexual dysfunction in women.
Nonetheless, diagnosing this condition can be challenging. Various factors influence libido, especially post-menopause when declining hormone levels cause several biological and medical changes. Physicians usually need to exclude other potential issues, ranging from relationship problems to mental health disorders, before prescribing any treatment.
The diagnosis itself is not without controversy; some psychologists argue that low sex drive should not be classified as a medical condition.
Addyi faced two rejections from the FDA before finally being approved in 2015, primarily due to its limited effectiveness and concerning side effects. This approval followed a lobbying effort from the company and advocates who framed the lack of treatment options for female libido as a matter of women’s rights.