Breakthrough in Cartilage Regeneration
A recent study led by Stanford Medicine has found that an injection designed to block a specific protein related to aging can reverse the natural loss of cartilage in the knee joints of older mice.
This treatment also appears to prevent arthritis that often follows knee injuries, such as ACL tears, which are common among athletes and those who engage in recreational activities. There’s even an oral version of this treatment undergoing clinical trials aimed at addressing age-related muscle weakness.
Interestingly, human knee tissue samples from replacement surgeries responded positively to the treatment, producing new and functional cartilage. This suggests that in the future, procedures like knee and hip replacements could become obsolete.
The approach focuses on osteoarthritis, a degenerative joint disease affecting roughly one in five adults in the U.S., incurring about $65 billion annually in healthcare costs. Currently, the only options available are preventative measures or joint replacements, as no existing drug can effectively slow down or reverse this condition.
The protein that the injection targets, known as 15-PGDH, acts as a major regulator of aging, increasing in quantity as we get older. These proteins, sometimes referred to as “gerozyms,” also contribute to the decline of tissue function, impacting muscle strength significantly among older mice.
In older animals, inhibiting 15-PGDH leads to an increase in muscle mass and endurance, while introducing more of this protein into younger mice results in muscle shrinkage. Regeneration in bone, nerve, and blood cells usually occurs thanks to the activity of tissue-specific stem cells. However, for cartilage, chondrocytes seem to change their gene expression to a more youthful state independently of stem cells.
“This represents a novel approach to regenerating adult tissue and offers considerable clinical hope for treating arthritis related to age or injury,” said Helen Blau, PhD, a microbiology and immunology professor. “We initially sought stem cells, but it’s clear they aren’t involved. It’s thrilling.”
Nidhi Bhutani, PhD, a co-author, added, “Millions experience joint pain and swelling as they age. This is a significant unmet medical need. Until now, there hasn’t been a treatment that directly targets the cause of cartilage loss. This gerozyme inhibitor demonstrates remarkable cartilage regeneration beyond what any other drug or intervention has achieved.”
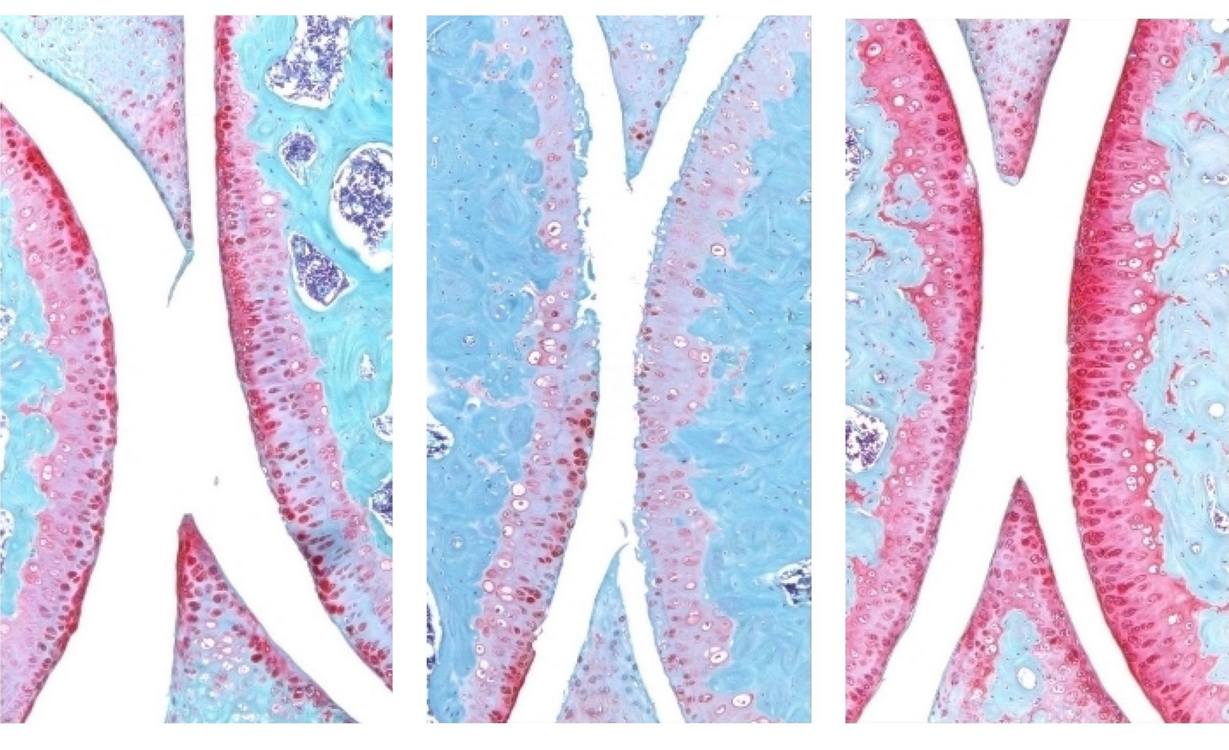
There are three primary types of cartilage in the human body. Hyaline cartilage, known for its smooth surface, usually covers joints such as the ankles and knees and is primarily affected by osteoarthritis. The condition can arise from aging, injury, or obesity, which leads chondrocytes to release inflammatory molecules and break down collagen—the main structural protein in cartilage. With collagen loss, the cartilage becomes thinner and softer, resulting in the inflammation and pain typical of the disease. Normally, articular cartilage has limited regenerative capacity, and efforts to identify stem or progenitor cells in wear areas have not succeeded.
Prior research from Blau’s lab indicated that a molecule called prostaglandin E2 is vital for muscle stem cell function. Since 15-PGDH breaks down prostaglandin E2, inhibiting its activity—or increasing prostaglandin E2 levels—supports the regeneration of various cell types in young mice.
Curious if 15-PGDH influenced aging cartilage, the researchers compared its levels in the knee cartilage of young and old mice, observing around a two-fold increase in older specimens. They then injected older animals with a small drug that inhibits 15-PGDH, starting with the abdomen and then directly into the knee joint. The results were impressive, with knee cartilage in older mice thickening and regenerating with genuine hyaline cartilage—much more functional than the fibrocartilage previously present.
“The extent of cartilage regeneration in aged mice surprised us,” Bhutani remarked. “The effect was astounding.”
Similar findings emerged in animals with knee injuries from ACL tears common in sports involving quick movements. Although these injuries can be surgically repaired, around 50% of individuals develop osteoarthritis in the affected joint within 15 years.
The researchers noted that a series of injections of the gerozyme inhibitor, given twice a week for four weeks post-injury, greatly lowered the likelihood of osteoarthritis developing in the treated mice. Those receiving the inhibitor also demonstrated more normal movement and placed greater weight on their injured leg compared to untreated ones.
“Interestingly, prostaglandin E2 is often associated with inflammation and pain,” Blau noted, “but our findings suggest that small increases in it—at normal biological levels—can actually promote regeneration.”
A deep dive into the chondrocytes of old versus young mice revealed that older chondrocytes exhibited more harmful genes associated with inflammation and the transformation of hyaline cartilage into unwanted bone, while showing reduced expression of genes vital to cartilage development.
In studies with human cartilage from osteoarthritis patients undergoing knee replacements, tissue treated with the 15-PGDH inhibitor for one week demonstrated lower levels of 15-PGDH and decreased markers of cartilage degradation. This treatment even sparked the regeneration of articular cartilage.
“The mechanism is remarkable and has altered our understanding of how tissue regeneration works,” Bhutani stated. “A significant number of pre-existing cells in cartilage adjust their gene expression patterns, and by focusing on these cells for regeneration, we may achieve a more substantial clinical impact.”
“Initial clinical trials of a 15-PGDH inhibitor aimed at muscle weakness have proven safe and effective in healthy participants,” Blau concluded. “We’re hopeful that trials will soon commence to explore its potential for cartilage regeneration. The prospect of regenerating existing cartilage and avoiding joint replacement is incredibly exciting.”