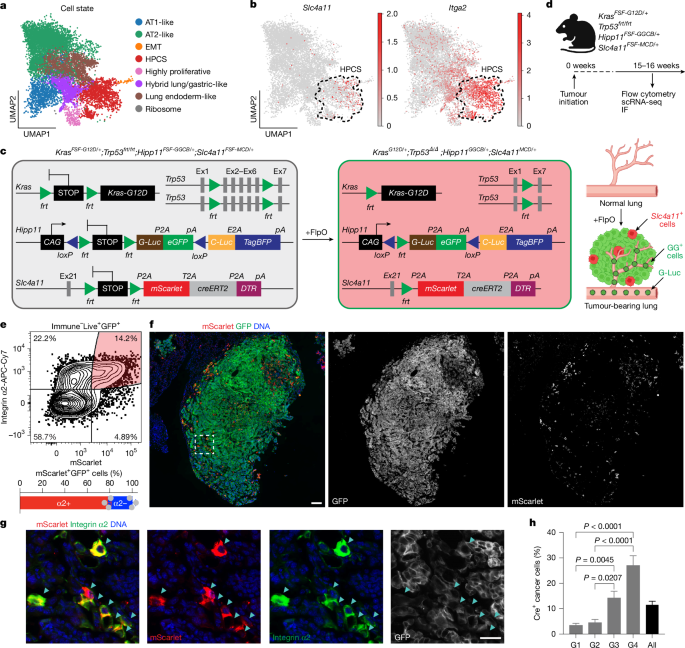
Mice
This study utilized various previously published genetically engineered mouse strains, including KrasLSL-G12D/+, Trp53flox/flox, KrasFSF-G12D/+, and others, with new reporter strains such as Hipp11FSF-GGCB and HopxFSF-MACD generated during the study. The mice had autochthonous KP lung tumors and were maintained in a mixed background of C57BL/6 × Sv129. For allotransplant studies, immunocompromised NSG mice were used as recipients. Throughout the study, all mice were kept under strict monitoring in a controlled environment.
Autochthonous and transplantation models of lung cancer
The autochthonous LUAD tumors were induced in specific mouse strains using various viral vectors, with doses ranging from 1 × 108 to 1 × 109 PFU. The experiments involved different methods of transplantation, including subcutaneous, orthotopic, and intravenous methods, using NSG mice as recipients for all treatments. The protocols adhered to relevant guidelines, and appropriate measures were taken for humane treatment and euthanasia when necessary.
Generation of donor vectors for embryonic stem cell targeting
The Slc4a11-FSF-MCD donor vector was developed using high-fidelity PCR and a specific cloning method. Similarly, the Hipp11-FSF-GGCB and Hopx-FSF-MACD donor vectors were created by amplifying corresponding regions and constructing homology-directed repair templates. Multiple techniques were applied to ensure successful vector assembly for subsequent targeting.
Validation of the Hipp11GGCB reporter
To verify the functionality of the GGCB cassette, AT2 cells from a chimeric mouse were transformed using lentiviral vectors. In these tests, the oncogenic KRAS(G12D) was activated, followed by the deletion of Trp53. After a 4-OHT stimulation, a switch in expression patterns was observed. Increased luciferase activity was noted over time following transformation, confirming the reporter’s activity across different experimental conditions.
Embryonic stem cell targeting, genotyping, and chimera generation
An embryonic stem cell line was generated by breeding specific mouse strains and subsequently culturing blastocysts on feeder layers. Genotyping was performed using PCR to confirm the desired genetic modifications in the ES cell lines.
Generation of genetically engineered reporter mouse strains
Chimeric mice were created by injecting genotype-verified ES cells into host embryos. These F0 mice were then crossed into specific backgrounds to establish genetically engineered strains for further experimentation.
Generation of LUAD reporter and lineage-tracing cell lines
The development of Slc4a11MACD/+;KP LUAD reporter cell lines involved co-transfection with donor vectors targeting Slc4a11. The resulting drug-resistant clones were selected and validated through genotyping. Additional cell lines were derived from organoid cultures and characterized for lineage tracing.
Dissociation of LUADs and lung tissue
To isolate cells, indicated tissues were processed through a series of enzymatic dissociations and filtration steps. The resulting cell populations were then used for various analyses aimed at understanding the tumor and normal lung cell environment.
Flow cytometry analysis and FACS
Prepared cells were subjected to flow cytometry to analyze specific markers. Sorting was performed to isolate desired cell populations for downstream applications, keeping in mind the need for precise identification based on surface markers.
Alveolar organoid culture and ex vivo transformation protocol
FACS-purified AT2 cells were transformed through lentiviral methods and cultured in organoid systems. The protocol involved mixing transformed cells with primary cells and using specialized culture media to maintain their growth over periods, with various analyses conducted to monitor changes over time.
IF and immunohistochemistry
The tissues were prepared through fixation and embedding before performing immunofluorescence or histological staining. Sufficient detail in procedures ensured accurate analysis of both tissue morphology and specific protein expression, leveraging various antibodies for detection.
In vivo EdU/BrdU dual labelling and imaging
Sequential labeling techniques were used to assess cellular proliferation in lung tissues, with subsequent histological preparations ensuring the integrity of observations made during imaging phases.
In situ hybridization
In situ hybridization was performed on FFPE tissues to analyze mRNA expression, utilizing commercially available kits. The probes and conditions were carefully selected to maximize outcomes relevant to the study’s hypotheses.
AkaLuc in vivo bioluminescence imaging
Mice with established LUAD tumors underwent bioluminescent imaging after substrate injections, with the goal of quantitatively measuring tumor activity across various experimental setups.
Statistics and reproducibility
Statistical analyses were conducted using appropriate tests, with a focus on ensuring repeatability and significance in findings. Experimental designs were rigorously controlled, allowing for robust comparisons among groups.
Reporting summary
Further information regarding the study’s design and outcomes is available in the supplementary materials.