Engineered Immune Cells Target Alzheimer’s Protein Plaques
Recent research shows that engineered immune cells can successfully target and reduce the toxic protein plaques associated with Alzheimer’s disease. This issue is becoming increasingly significant within the Department of Veterans Affairs medical system.
The study, published on February 9 in the Proceedings of the National Academy of Sciences, was conducted by researchers at Washington University School of Medicine in St. Louis, Missouri, who are affiliated with the VA St. Louis Health Care System. This marks the first application of CAR-T cell therapy—a method originally designed for treating cancer—toward addressing a neurodegenerative disease.
Importance for Veterans
According to estimates from the VA’s Veterans Health Administration, about 10% of VA patients aged 65 and older are diagnosed with dementia. Projections from the VA’s Office of Policy and Planning suggest that the number of affected patients is expected to rise by 22% between 2020 and 2033, increasing from around 276,000 to 335,000.
Interestingly, over half of all VA patients are in this age group. Moreover, veterans face unique risk factors compared to the general populace: longitudinal studies indicate that those with post-traumatic stress disorder (PTSD) are 50% to 60% more likely to develop dementia, and the risk linked to traumatic brain injuries could be even higher. A January 2026 study highlighted that veterans exhibit a higher prevalence of nearly every modifiable dementia risk factor, such as diabetes, chronic pain, depression, and sleep disturbances.
Findings from the VA’s Million Veteran Program reveal that PTSD and traumatic brain injury (TBI) not only coexist with various genetic risk factors for Alzheimer’s but may also exacerbate an individual’s biological predisposition to the disease. As the post-9/11 veteran generation ages and enters a critical risk phase for dementia, the demand for innovative treatment options is becoming urgent.
Understanding CAR-T Cells and Study Methods
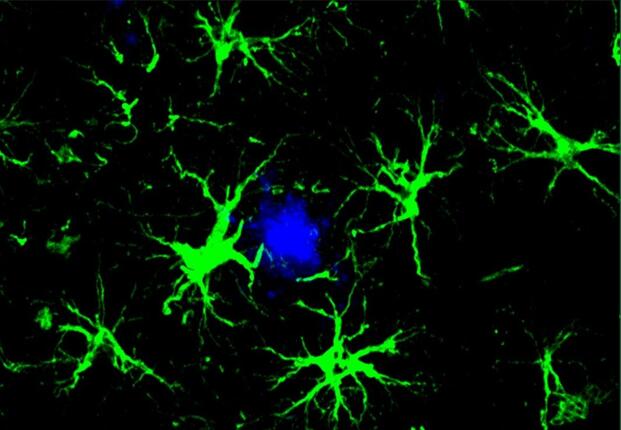
The CAR-T cell therapy works by extracting a patient’s own T cells—an immune cell type—and genetically modifying them to target specific issues. This process has been revolutionary for certain blood cancer treatments. The team at WashU, led by neuroimmunologist Dr. Jonathan Kipnis, adapted this technique for Alzheimer’s by modifying T cells to identify amyloid beta, a protein that contributes to the formation of dementia-related plaques.
The researchers took T cells from healthy mice, altered them to specifically target amyloid beta, and injected these into six-month-old mice that had been genetically engineered to develop Alzheimer’s-like conditions. The mice received three injections over a span of ten days, after which a notable reduction in amyloid plaques was observed in the treated group compared to controls that received unmodified T cells. Additionally, the brains of the treated mice displayed less activation of microglia and astrocytes, which are linked with the neuroinflammation related to Alzheimer’s.
Next Steps in Research
This study is still in its infancy, focusing on mice rather than humans, and the researchers recognize that much work lies ahead to evaluate safety, dosage, and the feasibility of applying this approach to the human brain, which is considerably more complex.
Dr. Kipnis has indicated that they intend to investigate how the engineered cells can further enhance brain health beyond just reducing plaques. There are also plans to assess this methodology in mouse models for other neurodegenerative diseases like ALS and Parkinson’s.
This research builds on Kipnis’s prior work uncovering the brain’s meningeal lymphatic system and his findings showing that T cells possessing neuroprotective traits can promote spinal cord recovery.
For veterans dealing with cognitive challenges from TBI or facing the early stages of Alzheimer’s, this research might be years away from clinical applications. Nevertheless, it contributes to the expanding field of research from the WashU-VA St. Louis collaboration, exploring the immune system’s potential to aid brain health.