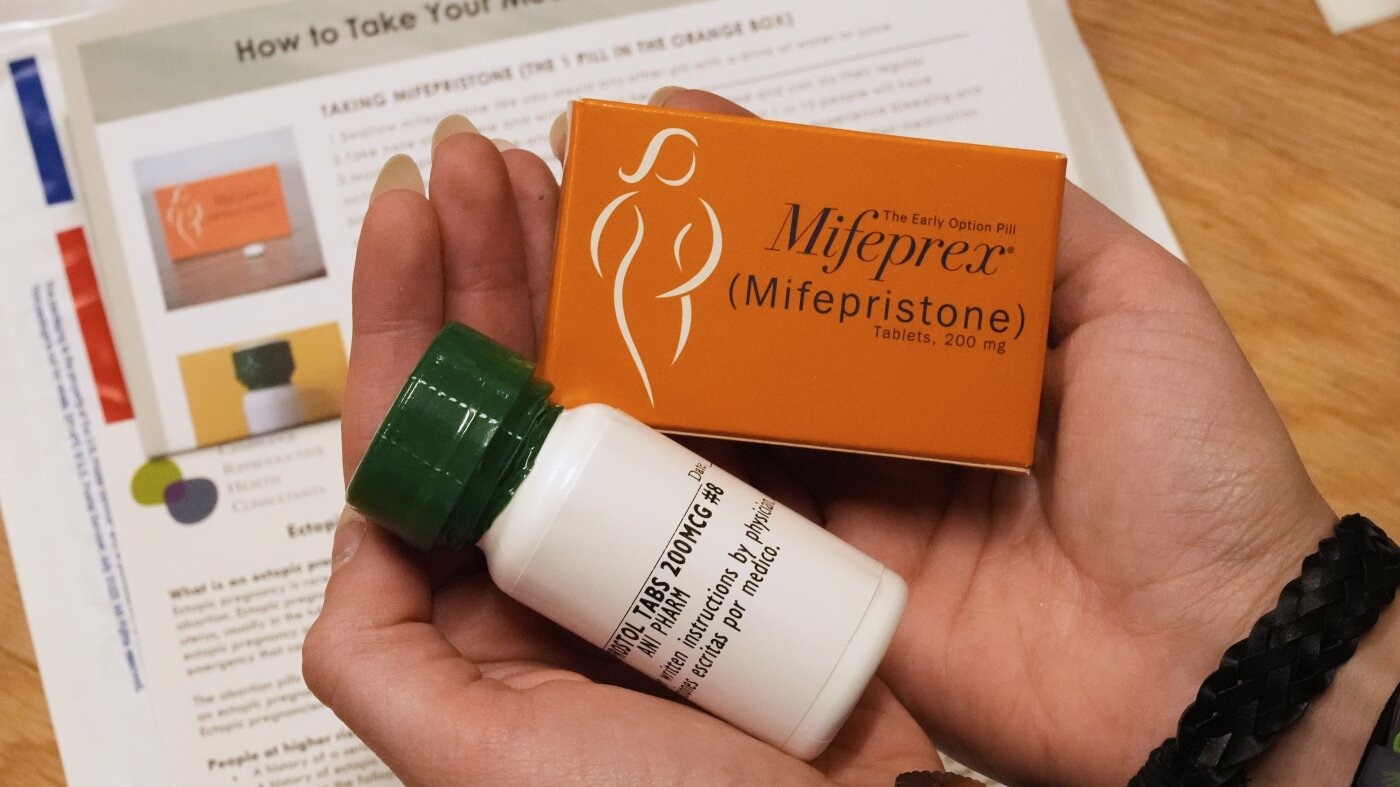
Picture this: You’re a few weeks into your pregnancy and you decide to have an abortion. You walk into a pharmacy, grab a box labeled “medication abortion kit,” pay for it, and leave to end your early pregnancy at home.
Dr. Daniel Grossman, who is part of a research team that just released a study in JAMA Internal Medicine, said, “It’s time that the general public understands that this could be a reality.”
Right now, over-the-counter abortion medication doesn’t exist. Grossman mentioned that he isn’t aware of any applications with the Food and Drug Administration (FDA) seeking to sell abortion medication without a prescription. To complicate things, over a dozen states have imposed strict bans on any form of abortion, and some are even pursuing legal action to make medication access even harder.
Grossman, who directs Advancing New Standards in Reproductive Health at the University of California, San Francisco, highlighted that there’s a lot of talk surrounding evidence-based restrictions regarding medication abortion. He points to decades of research that confirm the safety and effectiveness of the two primary drugs used for medication abortion. “It’s exciting to see science steering us in a new direction,” he noted, emphasizing that access could potentially be expanded.
The study design
In this study, researchers surveyed 168 individuals who were waiting to consult with a clinician about getting medication abortion. Grossman explains that if they were interested in participating, they were shown a box that resembled potential packaging for an over-the-counter abortion kit. They named this prototype “MiMi,” after the medications mifepristone and misoprostol. Creating the design took considerable time, Grossman added.
After reviewing the information on the box, participants assessed their own eligibility for medication. Researchers then compared these self-assessments with the evaluations made by clinicians later on.
The results showed that participants were quite accurate in evaluating their own eligibility; Grossman said nearly 88% had matching responses with clinicians—either both found them eligible or both deemed them ineligible.
However, he cautioned that the study sample was small and the findings may not be broadly applicable, stating, “This isn’t going to be the definitive study to sway the FDA, but it suggests a direction worth exploring further.”
Growing evidence
A commentary accompanying the study in JAMA Internal Medicine highlighted the increasing evidence advocating for over-the-counter access to medication abortion. Dr. Sonya Borrero from the University of Pittsburgh pointed out, though, that actual decision-making at the FDA may be swayed by the political climate surrounding reproductive health—essentially suggesting that scientific evidence might struggle against ideological pressures.
Julie Maslowsky, a psychologist and population health scientist at the University of Michigan, found the study’s results unsurprising. She remarked that the situation they’re investigating isn’t far removed from what many already experience in clinical settings. Many patients are engaging in telemedicine, receiving comprehensive information before their appointment, and ultimately using the medications at home.
She believes the medications meet safety and efficacy criteria for the FDA to consider an over-the-counter option, reflecting only a slight change from current availability in states where abortion is permitted.
If over-the-counter medication abortion seems far-fetched, she noted that it perhaps reflects the stigma surrounding reproductive health. She emphasized that the drugs work within the body much like other medications approved for over-the-counter sales.
The political moment
As of now, there’s no over-the-counter abortion kit available, and the current political environment makes that hard to envision.
During recent times, President Trump has seemed less inclined to act on abortion issues in his second term, but some Congressional Republicans have intensified their opposition. For example, Senator Josh Hawley has proposed legislation to revoke full approval of mifepristone, while Senator Bill Cassidy has initiated an investigation into the drug manufacturers involved. In a recent Senate hearing, Cassidy expressed concern about the current state of abortion pill access and safety.
Louisiana has even classified mifepristone as a controlled substance, leading to criminal charges against a physician who prescribed it via telemedicine. There’s currently a court case aiming to impose an in-person appointment requirement for mifepristone, which could diminish nationwide telehealth access to this medication.
An ongoing safety review by the FDA for mifepristone has been sparked by anti-abortion activists. The agency’s updated FAQ from February noted its commitment to conducting thorough scientific research.
Given the political landscape, Grossman suggested that now might not be the best time to approach the FDA for over-the-counter medication approval.
“A long road”
Looking ahead, Grossman drew parallels with the FDA’s long journey toward approving an over-the-counter birth control pill. He pointed out that initial groundwork involved research to demonstrate public interest in such access, bolstering pharmaceutical companies’ interest and paving the way for FDA discussions.
For medication abortion to transition to over-the-counter availability, Grossman notes, a pharmaceutical company would need to conduct substantive practical use studies—essentially making the drug accessible, tracking usage, and evaluating efficacy, which are often complex and costly endeavors.