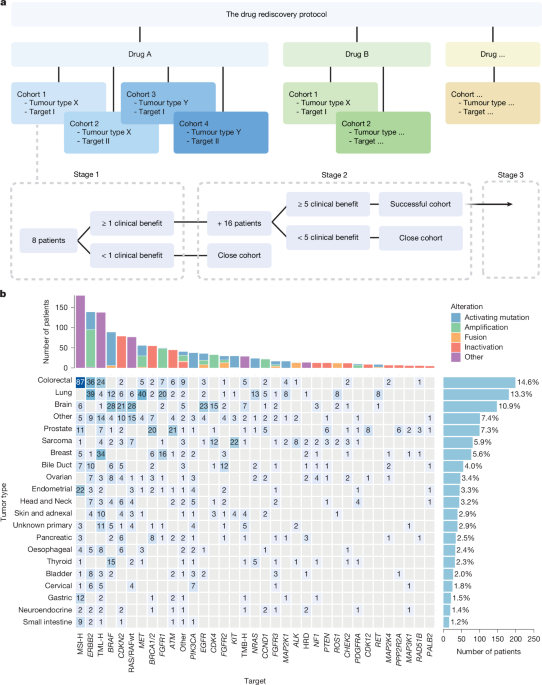
Insights into the Drug Rediscovery Protocol (DRUP)
Over the last nine years, the Drug Rediscovery Protocol (DRUP) has given hope to more than 1,600 patients suffering from advanced cancers that had no standard treatment options left. Notably, around 39.1% of these patients had rare cancers, which typically offer very few histological treatment choices. They appeared to gain just as much from the precision-based approach.
In the initial treatment stages, 34.9% of participants experienced some clinical advantage, while 15.7% showed an objective response. Interestingly, only 7.0% of these were considered exceptional responders. The Clinical Benefit Rate (CBR) and Objective Response Rate (ORR) for DRUP have been stable throughout the research period, aligning with findings from other precision oncology trials.
It’s important to note that off-label drugs are often prescribed outside of clinical trials, lacking the same level of oversight, which can present risks. Many patients, already in a fragile state after standard treatments, face potential serious side effects. In fact, in DRUP, 28.4% of the patients experienced grade 3 or higher treatment-related adverse events. On top of that, the costs associated with high drug prices and inconsistent reimbursements can put a financial strain on healthcare systems.
This scenario has led to calls for stricter regulations on off-label use, with structured programs like DRUP, TAPUR, and CAPTUR. These initiatives enhance controlled access to treatment while simultaneously generating valuable data. They not only provide options for rare cancers but also help in tracking patient outcomes and identifying both successes and failures.
Despite the modest overall activity, DRUP has also showcased significant potential benefits—numerous exceptional responders have been reported across various molecular subgroups. For example, patients with BRAF p.V600E-mutated brain tumors had notable responses. Additionally, one DRUP cohort collected enough evidence to secure full national reimbursement for its off-label use, suggesting that thorough evaluations can indeed broaden access when there’s clear efficacy.
When examining factors influencing patient benefit, several stood out. Firstly, the best-performing targets shared a solid biological rationale with prior clinical support. This is reflected in established classification systems that rank alterations by their actionability. For instance, BRAF p.V600E is currently at the top tier among actionable targets.
Secondly, patients with a smaller number of prior treatments tended to show better outcomes. This finding mirrors trends seen in other precision oncology trials, suggesting that administering treatments at earlier disease stages might be beneficial—but it should be weighed against standard treatment options that might also be available.
Thirdly, the context of the tissue type was also considered relevant. DRUP initially assumed tissue context impacted treatment response, but this led to many small cohorts that took time to enroll, complicating results. Only about 23.5% of drug-target combinations truly showed that tumor type made a difference in outcomes. This aligns with observations from other studies where tumor-agnostic responses differed across cancer types.
To ensure positive findings transition smoothly into clinical settings, DRUP includes a third-stage expansion strategy with personalized reimbursement models. However, despite meeting success criteria for numerous cohorts over the years, only one expansion has actually been completed. It seems some cohorts might have satisfied the preliminary criteria but lacked enough clinical significance to warrant further development.
Moreover, patent expiry issues often arise by the end of a cohort’s study, leading to diminished interest from pharmaceutical companies for future trials, and leaving patients without access to potentially beneficial therapies. Additionally, the restricted use of molecular diagnostics and the inherent rarity of some conditions have hindered progress. The DRUP framework has inspired similar trials across Europe, promoting better collaboration and data sharing.
There are limitations to consider when interpreting these findings. Many drugs studied in DRUP were developed before the current surge of next-gen therapies and may not encapsulate the full potential of modern precision oncology. Also, while all patients had exhausted standard treatments, there was still considerable variability in prior treatment lines, impacting the ability to detect clinical activity in some cases.
In conclusion, while oncologists often resort to off-label treatments based on scientific reasoning and hopes for effectiveness, the lack of a systematic framework to capture outcomes raises significant risks—both for patient safety and equity in access. DRUP serves as a case study highlighting the hurdles and possibilities of off-label drug use. Despite limited activity across the board, substantial benefits have emerged for specific groups, indicating that with robust evidence collection and treatment evaluation, off-label precision medicine can lead to meaningful patient outcomes.