Recommendations
This section outlines guidance for health professionals, spanning from the initial evaluation to the final stages of nPEP therapy. It addresses when nPEP should be administered, emphasizing rapid initiation, detailing preferred and alternative regimens, and discussing necessary laboratory tests and follow-ups. Additionally, it covers transitioning from nPEP to PrEP for ongoing prevention.
nPEP Indications
nPEP is appropriate for reducing the risk of HIV from high-risk exposures. Evaluating the risk involves considering factors like the HIV status of the source, their viral load, and the nature of the exposure.
Recommendations for Indications
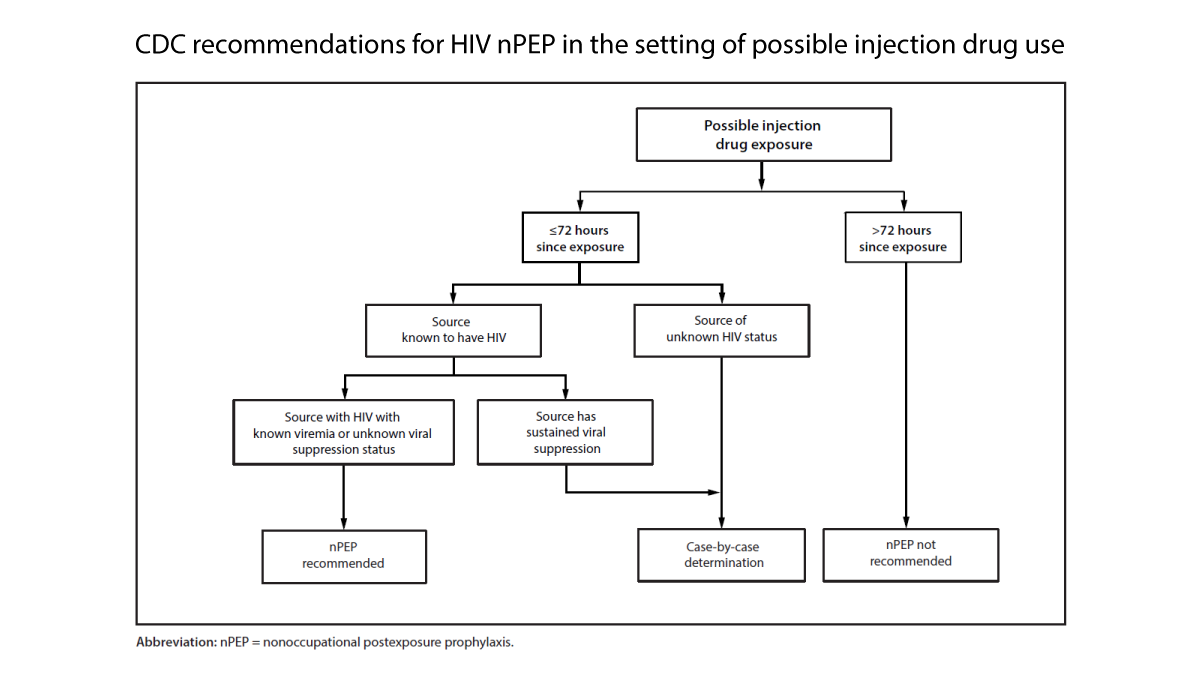
- Administer nPEP if exposure occurred within 72 hours and there’s a significant risk of HIV transmission without evidence of the source’s viral suppression.
- Assess on a case-by-case basis if exposure occurred within 72 hours and HIV status of the source is unknown.
- NPEP is not advised if the exposure poses no significant risk.
- Discontinue nPEP if the source is determined not to have HIV.
Rationale for Indications
For more specific scenarios regarding exposure and additional resources, decision algorithms can guide professionals. Although comprehensive data relating to the benefits of nPEP versus risks is limited, newer antiretroviral medications are safer and more tolerable.
HIV Status of the Source
If a source’s HIV status is unclear, clinicians should still proceed with nPEP, initiating treatment as quickly as possible. Testing of the source for HIV is recommended when feasible. If the source is confirmed to have HIV, information regarding their viral load is crucial for managing the exposed person’s care.
nPEP and PrEP Considerations
For individuals on PrEP, adherence is critical. Studies suggest substantial efficacy regarding HIV risk reduction based on dosing patterns. Any non-adherence should prompt consideration of nPEP for new exposures.
Timing of HIV nPEP Initiation
HIV exposure is an urgent matter, and nPEP should begin immediately, ideally within 72 hours, to maximize effectiveness.
Timing Initiation Recommendations
- Initiate nPEP as soon as possible within 72 hours following exposure.
Rationale for Timing
Research highlights decreased effectiveness of nPEP the longer the initiation is delayed beyond 72 hours. However, some experts believe newer ARVs may still be effective beyond this time frame, although there are no definitive studies to support that.
HIV nPEP Regimens
A 28-day course of nPEP is recommended for those who seek care shortly after non-occupational exposures to potentially infectious body fluids. The regimen should provide strong efficacy while remaining user-friendly to encourage adherence.
Regimen Recommendations
- Conduct a clinical assessment prior to prescribing nPEP, considering medical history and potential drug interactions.
- The advised duration for nPEP is 28 days.
- Preferred regimens include:
- bictegravir/emtricitabine/tenofovir alafenamide OR
- dolutegravir with either tenofovir alafenamide or tenofovir disoproxil fumarate, combined with emtricitabine or lamivudine.
- Choose regimens tailored to the individual’s health status and potential drug interactions.
Rationale for Regimens
While rigorous clinical trials on nPEP efficacy are lacking, available evidence supports the use of three-drug regimens for optimal prevention of resistant HIV strains. Although some two-drug regimens exist, they are not recommended for nPEP without clear justification.
Preferred and Alternative Regimens
The optimal nPEP regimens include two NRTIs with a second-generation INSTI. Providers may consider regimens outside the standard recommendations based on specific patient circumstances, consulting when necessary.
Choosing an Initial Regimen
Choosing a regimen involves weighing existing health conditions and histories, which can impact both dosing and monitoring requirements.
Considerations for Special Conditions
Health status, such as renal function, can impact medication choice, requiring dosage adjustments and additional monitoring. Pregnancy and breastfeeding should not deter the use of nPEP, although careful consideration of safety data for drug choice is critical.
Adverse Events and Drug Interactions
Interactions between nPEP and other medications can significantly impact therapy effectiveness and safety. A detailed medication history should always be obtained before prescribing. Some ARVs are known to have tolerability issues, necessitating proactive management strategies for potential side effects.
Source History and Resistance
If the source’s HIV status is known, the nPEP regimen can be tailored. However, if this information is delayed, treatment should not be postponed. Understanding the potential for resistance is important if initiating nPEP with knowledge of the source’s treatment history.
Follow-Up Care and Support
Follow-up visits post-nPEP initiation ensure medications are tolerated and track laboratory results. A follow-up plan should be established at the initial visit to reinforce ongoing care and engagement with healthcare providers.
Laboratory Testing and Follow-Up
Laboratory tests at nPEP initiation are crucial to exclude existing HIV infection and assess other health metrics. Follow-up testing is recommended to check for new infections post-treatment.
Recommendations for Laboratory Follow-Up
- Conduct initial HIV testing for all individuals undergoing nPEP assessment.
- Perform additional lab tests based on exposure circumstances.
- Interim and follow-up HIV tests should occur at specified intervals.
Initial Testing Objectives
Initial testing seeks to confirm HIV status and should not delay treatment initiation. Information about the source’s status is beneficial but not essential.
Follow-Up Testing Protocols
Follow-up tests should coincide with the completion of nPEP. Ideally, tests should detect any potential seroconversion, remaining vigilant for any signs of acute HIV infection.
Other Laboratory Testing Considerations
Broad testing that includes kidney and liver function, along with consideration for STIs, is warranted. If facing adverse effects, monitoring and testing should adapt to patient needs and outcomes during nPEP.
Transitioning to PrEP After nPEP
Face-to-face discussions about considering PrEP after completing nPEP can support ongoing prevention for those at continued risk.
Transitioning Recommendations
- Consider immediate transition from nPEP to PrEP after completion, particularly for those with ongoing risk.