New data indicates that adverse reactions related to popular weight-loss medications, such as Ozempic and Wegovy, could increase by over 350% within a year.
According to figures from the UK’s drug regulatory agency, if current trends persist, 2024 may see approximately 7,200 reported adverse reactions, a significant rise compared to 1,592 reported in 2023—about four and a half times lower than the anticipated increase.
However, due to delays in data collection, available figures only represent the period from January to May last year, amounting to 2,780 cases, with 281 considered serious.
The types of reactions reported range from severe gastrointestinal issues, such as stomach paralysis and bowel obstruction, to less extreme complaints like vomiting and bloating.
This data is compiled by the Medicines and Healthcare products Regulatory Agency (MHRA) from reports submitted by patients and healthcare professionals suspecting a drug’s role in their health issues.
It’s important to note that these reports have not been independently verified. In the United States, over 200 deaths have been associated with Ozempic and competing treatments, though a direct causal link hasn’t been established.
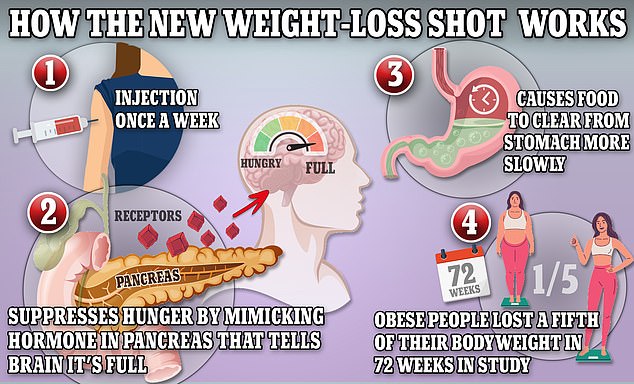
In the UK, there have been 82 reported fatalities related to these drugs, with no confirmed links to their use. Ozempic and Wegovy include the active ingredient semaglutide, which mimics a hormone that helps regulate appetite and digestion.
This process helps users feel fuller and decreases sugar production by the liver, promoting the burning of stored fat for energy.
Since its approval for weight loss in November 2023, Mounjaro, a drug with a similar mechanism to semaglutide, has already been linked to 209 adverse reactions, including one fatal incident.
The most frequently reported complaints across all these drugs have been fatigue and headaches, while less common side effects include menstrual bleeding, joint pain, and abnormal heart rhythms.
One user, Karen Coe, 59, reported harrowing side effects shortly after starting Mounjaro to control her type 2 diabetes. Within three days, she experienced severe headaches and dizziness, followed by intense stomach cramps.
On one occasion, her husband had to call an ambulance due to her collapsing. Despite being told she was fine and sent home, she endured painful cramps and blood in her stool over the next 24 hours.
A week later, Ms. Coe was hospitalized due to massive blood clots, and although doctors didn’t definitively connect her condition to Mounjaro, they suspected the drug had caused initial symptoms.
Ms. Coe advises others to carefully weigh the risks and side effects—such as nausea and abdominal discomfort—before trying these drugs. She emphasized, “It can cause severe reactions, and people should really think carefully.”
In response, Eli Lilly, the manufacturer of Mounjaro, stated that patient safety is their primary concern. They monitor and evaluate safety information for all their medications and encourage patients to consult healthcare professionals regarding any experienced side effects.
The information leaflet for Mounjaro warns about various common gastrointestinal side effects. However, the yellow card website, in partnership with the MHRA, points out that the adverse reactions reported have not yet been conclusively linked to the drug.
Here’s a brief summary of adverse reaction reports over the years related to semaglutide:
- 2019: 114 reported
- 2020: 144 reported
- 2021: 336 reported
- 2022: 534 reported
- 2023: 1,592 reported
- 2024 (up to May 19): 2,780 reported