Discovery Links Oral Bacteria to Parkinson’s Disease via Gut-Brain Connection
Researchers from Korea have found significant evidence suggesting that oral bacteria may influence neurons in the brain, potentially leading to Parkinson’s disease when they colonize the gut.
This study, spearheaded by Professor Ara Koh and doctoral candidate Hyunji Park from POSTECH’s Department of Life Sciences, involved collaboration with Professor Yunjong Lee and doctoral candidate Jiwon Cheon from Sungkyunkwan University School of Medicine, alongside Professor Han-Joon Kim from Seoul National University College of Medicine.
The team outlined how metabolites generated by oral bacteria in the gut could initiate the onset of Parkinson’s disease. Their findings were shared in a recent online publication in Nature Communications.
Parkinson’s disease is a significant neurological condition, marked by symptoms like tremors, rigidity, and slowed movements, affecting roughly 1–2% of people over 65 worldwide. It’s one of the most prevalent age-related brain disorders. While prior research indicated that the gut microbiomes of Parkinson’s patients differ from those of healthy individuals, the specific bacteria and metabolites involved weren’t well understood.
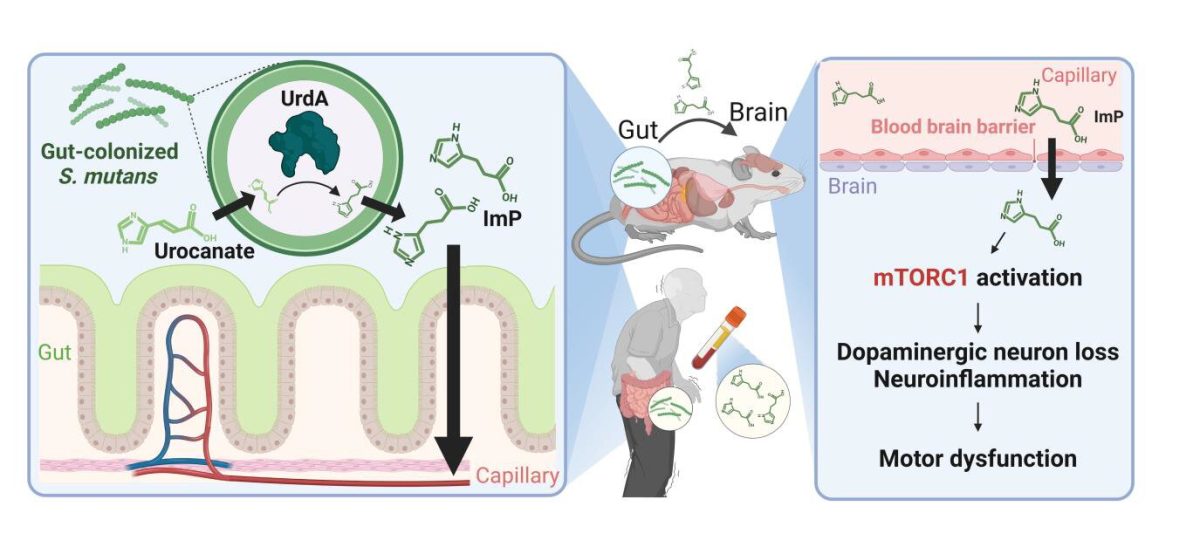
The researchers discovered a notable increase in Streptococcus mutans, an oral bacterium linked to dental caries, within the gut microbiomes of Parkinson’s sufferers.
More crucially, S. mutans produces the enzyme urocanate reductase (UrdA), along with its metabolite imidazole propionate (ImP), both of which were found in elevated concentrations in the gut and blood of affected patients. There were indications that ImP could travel through systemic circulation, reaching the brain and contributing to the degeneration of dopaminergic neurons.
Using mouse models, the research team introduced S. mutans into the gut or genetically modified E. coli to express UrdA.
The result? The mice exhibited increased ImP levels in their blood and brain tissue, along with classic Parkinson’s disease symptoms: loss of dopaminergic neurons, increased neuroinflammation, impaired motor function, and a higher aggregation of alpha-synuclein, a protein crucial to the disease’s progression.
Further tests indicated that these changes relied on activating the signaling protein complex mTORC1. Mice treated with an mTORC1 inhibitor saw a significant reduction in neuroinflammation, neuronal loss, alpha-synuclein aggregation, and motor dysfunction.
This implies that focusing on the oral-gut microbiome and its metabolites could lead to new treatment strategies for Parkinson’s disease.
Professor Ara Koh remarked, “Our study enhances understanding of how oral microbes in the gut can impact the brain and play a role in Parkinson’s disease development. It underscores the potential of addressing the gut microbiota as a therapeutic approach, pointing toward a new pathway for treating Parkinson’s.”
More information: Hyunji Park et al, Gut microbial production of imidazole propionate drives Parkinson’s pathologies, Nature Communications (2025). DOI: 10.1038/s41467-025-63473-4