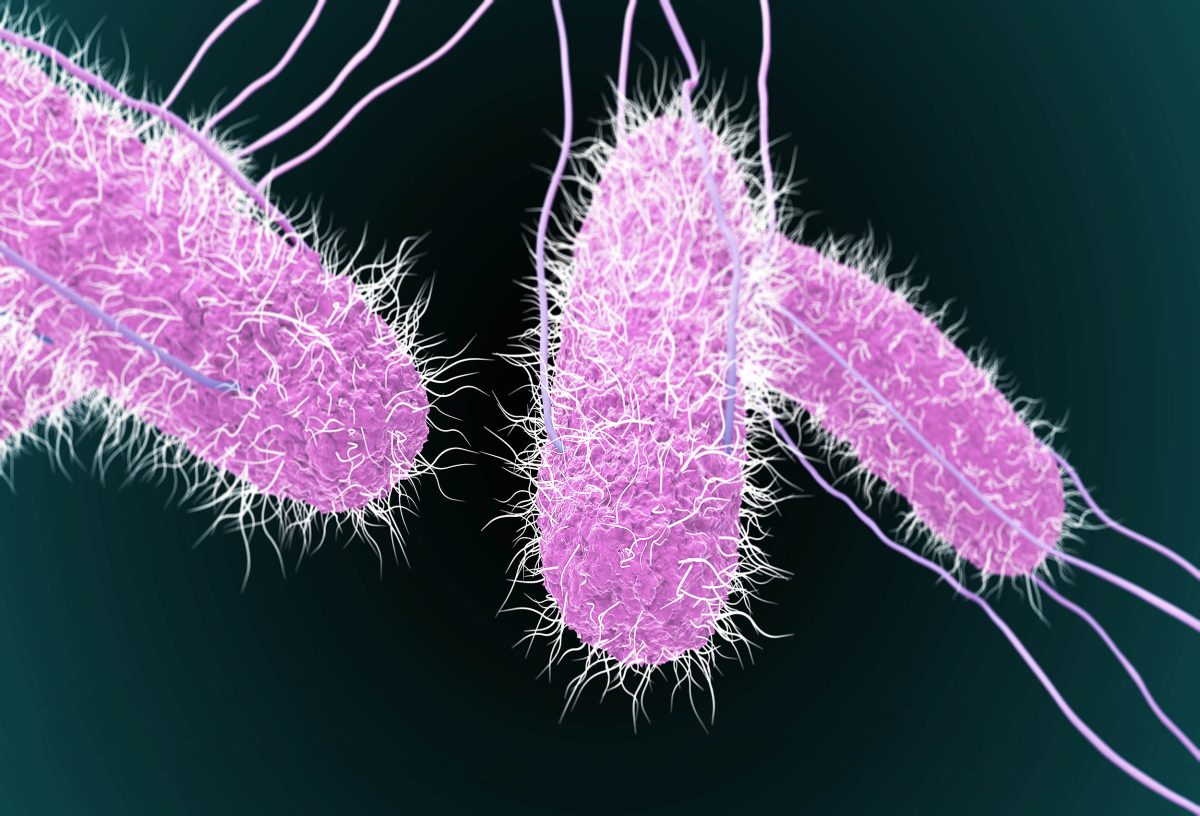
Understanding Salmonella Dublin
When people think of Salmonella poisoning, they often picture severe stomach issues from undercooked chicken or eggs. But there’s another strain, Salmonella Dublin, mostly linked to cattle, and it can lead to even more serious health problems.
Infections from this particular bacteria have been resulting in increased bloodstream infections, longer hospital stays, and a greater risk of severe outcomes. This trend of rising cases has raised concerns among health professionals.
How does Salmonella Dublin get transmitted? Common sources include unpasteurized milk and undercooked beef, along with contact with sick animals. This isn’t just a farm issue; it has broader public health implications, as the effects of cattle-related infections can spill over into the wider community.
Humans, Cattle, and Salmonella Dublin
This has led scientists to ponder an important question: Are the Salmonella Dublin strains found in cattle, humans, and agricultural environments similarly related in the U.S.?
The answers provide insights into prevention strategies and antibiotic use. If the strains are closely related across different hosts, then a unified approach is necessary—one that connects animal health, human health, and environmental monitoring. This is often referred to as the “One Health” concept.
This idea goes beyond a mere slogan. If the same bacteria can move between different species and environments without significant changes, it’s clear that focusing on just one area of the supply chain won’t suffice. Solutions need to address the bacteria wherever it may go.
Research on Salmonella Dublin
Researchers collected samples of Salmonella Dublin from three sources: sick cattle, infected humans, and environmental sites related to farms or processing facilities. They sequenced the DNA from these samples to see how closely the strains are connected.
In genetic research, scientists look for small changes known as single nucleotide polymorphisms (SNPs). A smaller number of SNP differences typically indicates a recent common ancestor.
The objective was to determine if these strains clustered together or formed distinct groups, which would indicate movement between hosts rather than isolated incidents of disease.
Current Findings
The results revealed that Salmonella Dublin strains from cattle, humans, and the environment in the U.S. were very similar, often differing by just a few SNPs. A study published in Applied and Environmental Microbiology highlighted that despite some genetic variations across 2,150 strains, the bacteria generally maintained a high degree of similarity.
“This similarity suggests significant potential for cross-transmission,” stated Erika Ganda, the lead researcher and an associate professor focusing on food animal microbiomes at Penn State’s College of Agricultural Sciences. Ganda emphasized that efforts to control Salmonella Dublin should consider the interconnectedness of humans, animals, and the environment.
“The findings can inform surveillance efforts to track the bacteria and to shape intervention strategies, such as regulating antibiotic use in livestock, along with public health policies,” she added.
Salmonella Dublin’s Genetic Structure
The genetic analysis revealed a close-knit relationship among strains, lacking distant branches based on their origins or timings. This indicates ongoing interactions among animals, humans, and farming environments.
Such interconnected patterns are significant for planning in hospitals, developing farming policies, and making informed choices about food at home.
The team also investigated antimicrobial resistance in these strains. They didn’t just ascertain its presence; they analyzed the specific resistance genes and found notable differences based on the source of the strains.
This suggests that control strategies should be tailored to the unique resistance patterns in each environment. A universal antibiotic strategy may not be effective.
Using antibiotics responsibly means not just guessing, but involves systematic lab testing and clear guidelines to protect the drugs that are still effective.
Prevention Strategies
Preventive measures should start before individuals become ill. On farms, implementing biosecurity to keep new or sick animals from spreading infections is critical, along with thorough cleaning and disinfecting practices. In the food supply chain, basic precautions like pasteurizing milk and thoroughly cooking beef can go a long way.
In home kitchens, avoiding raw milk and using a meat thermometer are advisable. Ground beef should reach at least 160°F, while whole cuts, like steaks, should reach 145°F with resting time included.
For healthcare settings, rapid lab testing plays a crucial role in identifying ineffective antibiotics early on. This ensures timely and appropriate treatments, reserving more potent drugs for critical situations.
The Power of Genome Sequencing
Genome sequencing provides a detailed “map” of thousands of bacterial samples, enabling scientists to identify connections that older methods might overlook, including hidden correlations between agricultural sources and human infections.
This allows public health teams to hone in on the pathways causing the most harm. Targeted measures can be far more effective than broad, generic strategies, especially when resources and time are constrained.
Salmonella Dublin and Public Health
This isn’t just a cautionary tale or a sensationalized story; it’s a meticulously conducted scientific study backed by peer-reviewed research.
The U.S. Salmonella Dublin population appears closely linked and shows little variation as it spreads. This clarity is beneficial, as it outlines a collective responsibility and a long list of actions that need to be taken.
Authorities must enhance surveillance that connects animal, human, and environmental data, tailor antibiotic guidelines to address the specific resistance patterns found, and mitigate clear transmission routes through pasteurization, careful cooking, farm hygiene, and effective infection control in medical facilities.
In straightforward terms, this particular pathogen keeps coming up in cattle, humans, and surrounding areas with nearly identical genetic profiles. Thus, with a coordinated approach, there’s a better chance to rein in its spread before it becomes a significant public health crisis.
The full study can be found in the journal Applied and Environmental Microbiology.