A brain mapping study funded by the NIH reveals which cell types may be the first to be affected.
New research funded by the National Institutes of Health (NIH) suggests that Alzheimer’s disease may impact the brain in two distinct stages. Using advanced brain mapping technology, researchers propose that an initial, early phase unfolds gradually and silently, targeting only a small number of vulnerable cell types long before memory issues arise.
In contrast, the later phase is marked by widespread damage, aligning with the onset of symptoms and a rapid buildup of plaques, tangles, and other characteristic signs of Alzheimer’s.
“One of the challenges to diagnosing and treating Alzheimer’s is that much of the damage to the brain happens well before symptoms occur. The ability to detect these early changes means that, for the first time, we can see what is happening to a person’s brain during the earliest periods of the disease,” said Richard J. Hodes, M.D., director, NIH National Institute on Aging. “The results fundamentally alter scientists’ understanding of how Alzheimer’s harms the brain and will guide the development of new treatments for this devastating disorder.”
Cellular Analysis and Key Findings
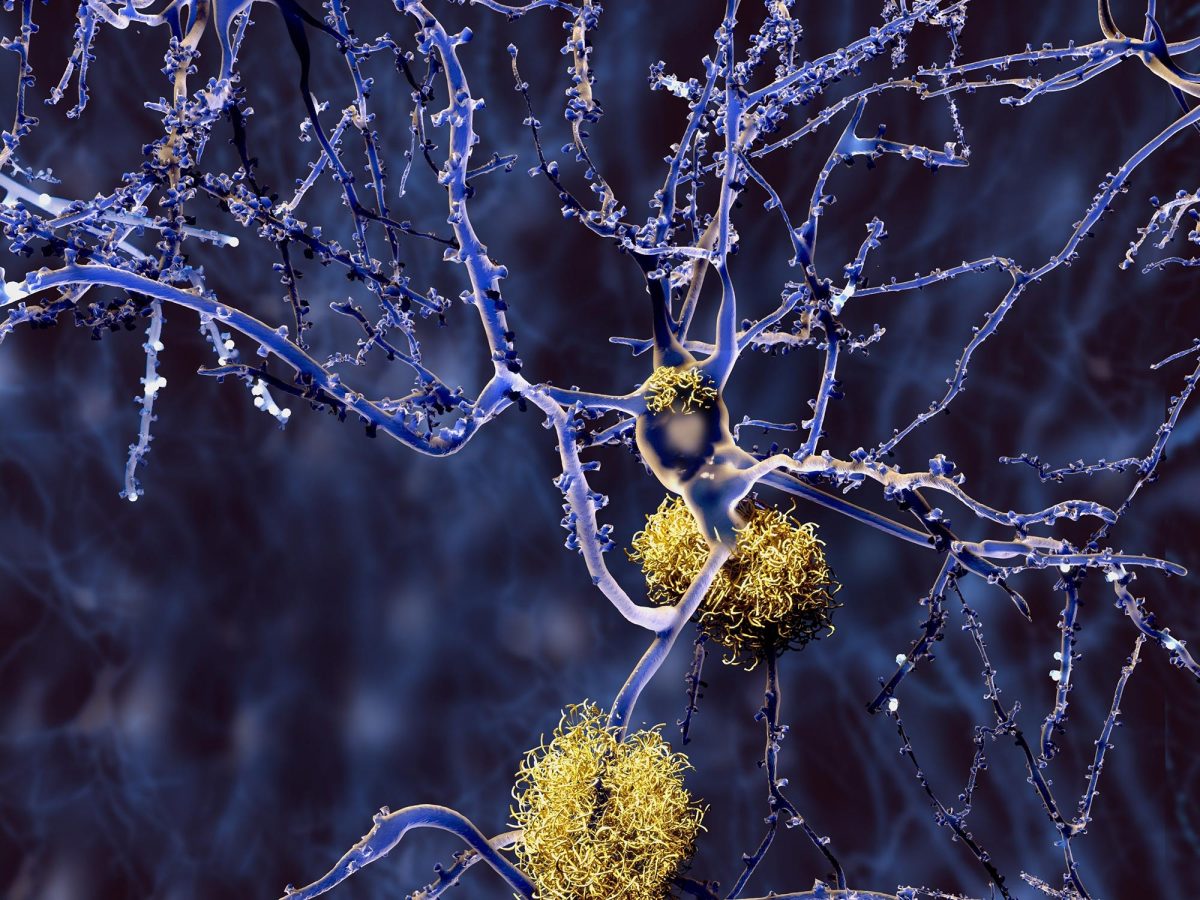
Scientists analyzed the brains of 84 people, and the results, published in Nature Neuroscience, suggest that damage to one type of cell, called an inhibitory neuron, during the early phase may trigger the neural circuit problems that underlie the disease. Additionally, the study confirmed previous findings about how Alzheimer’s damages the brain and identified many new changes that may happen during the disease.

Specifically, the scientists used advanced genetic analysis tools to study the cells of the middle temporal gyrus, a part of the brain that controls language, memory, and vision. The gyrus has been shown to be vulnerable to many of the changes traditionally seen during Alzheimer’s.
It is also a part of the brain that researchers have thoroughly mapped for control donors. By comparing control donor data with that from people who had Alzheimer’s, the scientists created a genetic and cellular timeline of what happens throughout the disease.
Rethinking Alzheimer’s Progression: A Two-Phase Model
Traditionally, studies have suggested that the damage caused by Alzheimer’s happens in several stages characterized by increasing levels of cell death, inflammation, and the accumulation of proteins in the form of plaques and tangles. In contrast, this study suggests that the disease changes the brain in two “epochs” — or phases — with many of the traditionally studied changes happening rapidly during the second phase. This coincides with the appearance of memory problems and other symptoms.

The results also suggest that the earliest changes happen gradually and “quietly” in the first phase before any symptoms appear. These changes include slow accumulation of plaques, activation of the brain’s immune system, damage to the cellular insulation that helps neurons send signals, and the death of cells called somatostatin (SST) inhibitory neurons.
The last finding was surprising to the researchers. Traditionally, scientists have thought that Alzheimer’s primarily damages excitatory neurons, which send activating neural signals to other cells. Inhibitory neurons send calming signals to other cells. The paper’s authors hypothesized how the loss of SST inhibitory neurons might trigger the changes to the brain’s neural circuitry that underlie the disease.
New Discoveries in Alzheimer’s Gene Research
Recently, a separate NIH-funded brain mapping study by researchers at MIT found that a gene called REELIN may be associated with the vulnerability of some neurons to Alzheimer’s. It also showed that star-shaped brain cells called astrocytes may provide resilience to or resist the harm caused by the disease.
Researchers analyzed brains that are part of the Seattle Alzheimer’s Disease Brain Cell Atlas (SEA-AD), which is designed to create a highly detailed map of the brain damage that occurs during the disease. The project was led by Mariano I. Gabitto, Ph.D., and Kyle J. Travaglini, Ph.D., from the Allen Institute, Seattle. The scientists used tools — developed as part of the NIH’s Brain Research Through Advancing Innovative Neurotechnologies® (BRAIN) Initiative – Cell Census Network (BICCN) — to study more than 3.4 million brain cells from donors who died at various stages of Alzheimer’s disease. Tissue samples were obtained from the Adult Changes in Thought study and the University of Washington Alzheimer’s Disease Research Center(link is external).
“This research demonstrates how powerful new technologies provided by the NIH’s BRAIN Initiative are changing the way we understand diseases like Alzheimer’s. With these tools, scientists were able to detect the earliest cellular changes to the brain to create a more complete picture of what happens over the entire course of the disease,” said John Ngai, Ph.D., director of The BRAIN Initiative®. “The new knowledge provided by this study may help scientists and drug developers around the world develop diagnostics and treatments targeted to specific stages of Alzheimer’s and other dementias.”
Reference: “Integrated multimodal cell atlas of Alzheimer’s disease” by Mariano I. Gabitto, Kyle J. Travaglini, Victoria M. Rachleff, Eitan S. Kaplan, Brian Long, Jeanelle Ariza, Yi Ding, Joseph T. Mahoney, Nick Dee, Jeff Goldy, Erica J. Melief, Anamika Agrawal, Omar Kana, Xingjian Zhen, Samuel T. Barlow, Krissy Brouner, Jazmin Campos, John Campos, Ambrose J. Carr, Tamara Casper, Rushil Chakrabarty, Michael Clark, Jonah Cool, Rachel Dalley, Martin Darvas, Song-Lin Ding, Tim Dolbeare, Tom Egdorf, Luke Esposito, Rebecca Ferrer, Lynn E. Fleckenstein, Rohan Gala, Amanda Gary, Emily Gelfand, Jessica Gloe, Nathan Guilford, Junitta Guzman, Daniel Hirschstein, Windy Ho, Madison Hupp, Tim Jarsky, Nelson Johansen, Brian E. Kalmbach, Lisa M. Keene, Sarah Khawand, Mitchell D. Kilgore, Amanda Kirkland, Michael Kunst, Brian R. Lee, Mckaila Leytze, Christine L. Mac Donald, Jocelin Malone, Zoe Maltzer, Naomi Martin, Rachel McCue, Delissa McMillen, Gonzalo Mena, Emma Meyerdierks, Kelly P. Meyers, Tyler Mollenkopf, Mark Montine, Amber L. Nolan, Julie K. Nyhus, Paul A. Olsen, Maiya Pacleb, Chelsea M. Pagan, Nicholas Peña, Trangthanh Pham, Christina Alice Pom, Nadia Postupna, Christine Rimorin, Augustin Ruiz, Giuseppe A. Saldi, Aimee M. Schantz, Nadiya V. Shapovalova, Staci A. Sorensen, Brian Staats, Matt Sullivan, Susan M. Sunkin, Carol Thompson, Michael Tieu, Jonathan T. Ting, Amy Torkelson, Tracy Tran, Nasmil J. Valera Cuevas, Sarah Walling-Bell, Ming-Qiang Wang, Jack Waters, Angela M. Wilson, Ming Xiao, David Haynor, Nicole M. Gatto, Suman Jayadev, Shoaib Mufti, Lydia Ng, Shubhabrata Mukherjee, Paul K. Crane, Caitlin S. Latimer, Boaz P. Levi, Kimberly A. Smith, Jennie L. Close, Jeremy A. Miller, Rebecca D. Hodge, Eric B. Larson, Thomas J. Grabowski, Michael Hawrylycz, C. Dirk Keene and Ed S. Lein, 14 October 2024, Nature Neuroscience.
DOI: 10.1038/s41593-024-01774-5
This study was funded by NIH grants: U19AG060909, P30AG066509, U19AG066567, U01AG006781. Additional funding was provided by the Nancy and Buster Alvord Endowment. The Rush University Alzheimer’s Disease Center, Chicago, Il, shared donor metadata from the Religious Orders Memory/Memory and Aging Project.
Researchers can obtain data from the SEA-AD study by going to the study’s website.