Searching for a Microbial Hero
I’m on a quest for a microbial savior. Not the kind of virus that makes us sick—I’m looking for one that can actually help us fight infections. You know the usual suspects: Covid, flu, norovirus, and so on. But there’s another type of virus that’s not interested in us at all; instead, it targets bacteria. These are known as bacteriophages, or simply phage.
Capturing these might open up new avenues for treating infections, especially those pesky superbugs that resist our current medications. So, how do you catch one of these viral fighters?
Surprisingly, it’s supposed to be fairly simple. The Phage Collection Project sent me some vials and gloves; my mission? Find some murky water, dip the vials in, and close them up tightly.
I figured a few muddy ponds and even some liquid from my worm bin would do the trick. But I needed something really dirty. So, I hesitated—after a trip to the bathroom, I left it for a couple of hours before donning a glove and collecting the sample. I assure you, I followed strict hygiene protocols and washed my hands thoroughly afterward!
Three days later, I was off to the University of Southampton to see what I had caught. “The samples were a bit murky,” said phage scientist Michelle Lin, as we suited up in lab coats and gloves to enter the microbiology lab.
We pulled my samples from the fridge. They looked clearer now, filtered from the debris. “Don’t worry; this is part of the process,” Michelle reassured me with a laugh.

The next steps involved filtering the samples and then feeding them a nutrient cocktail of what I can only assume is bacteria buffet to help them multiply. Here comes the fascinating part—discovering useful phages. The lab has been collaborating with a local hospital to acquire bacteria from patients battling tough infections.
At one point, Michelle pulled out a petri dish containing bacteria from a patient struggling with recurring urinary tract infections. To my astonishment, one of the phages I’d collected from my “special” sample was effective at killing that infection in the lab!
“You can see where the phages have infected the bacteria; there are clear zones where the bacteria aren’t growing, meaning they’ve been killed off,” Michelle explained.
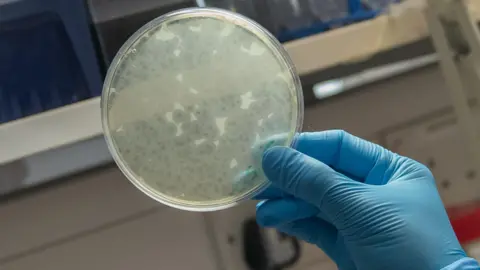
The petri dish had a leopard-print appearance, marking the areas where the phages had taken out the bacteria that conventional treatments struggled with. “As strange as it sounds, cheers to the toilet sample,” Michelle said with genuine excitement.
When given the opportunity to name the phage, I went for “Gallagher-phage.” Michelle seemed to think that was a pretty cool name.
Now, the question lingered—could this phage actually be used on a patient? “Yes, and I certainly hope so,” Dr. Franklin Nobrega chimed in while we examined electron microscope images of my phage.

“Your phage, in just 24 hours, has reached a high concentration and exhibited impressive killing capability. This is incredibly promising for future treatments,” Dr. Nobrega noted enthusiastically.
Phages are quite fascinating—they resemble moon landers, with spindly legs that allow them to latch onto their bacterial targets. Once attached, they hijack the bacteria, turning it into a factory for more phages—these then burst out, killing the host bacterium in the process.

There are both advantages and challenges with phages. They multiply on their own, so you don’t necessarily need to keep dosing like with conventional antibiotics. However, they are also really picky about which bacteria they target. This means finding the right phage for a specific bacterial strain can be trickier than just using antibiotics, which tend to wipe out a broad spectrum of bacteria.
Dr. Nobrega mentioned that phage therapy could be particularly effective for infected wounds, as well as administrable through nebulizers for lung infections or even urinary tract issues, which, notably, is their current focus.
History and Future of Phages
Though the concept of phage therapy may sound cutting-edge, it has roots that stretch back over a century to the discoveries made by Felix d’Hérelle and Frederick Twort in the 1910s. Even as late as the 1940s, there was significant interest in developing phage-based treatments in Western medicine. But then antibiotics came along and overshadowed phage therapy’s potential.
“Antibiotics worked so effectively that many people thought, ‘why bother?'” Dr. Nobrega noted.
Nonetheless, research into phage therapy has persisted, particularly in places like Georgia. Although successes have been documented, the breadth of clinical research isn’t comparable to that of pharmaceuticals.
Ironically, as antibiotic efficacy declines, interest in phage therapy is being rekindled. It’s predicted that over a million people die annually due to resistant infections, and that number could escalate to ten million by 2050—a looming “antibiotic apocalypse” that could compromise many aspects of modern medicine.
As Prof. Paul Elkington pointed out, these trends are alarming, and we are witnessing them now rather than later. The possibility of phage therapy moving from lab to clinical use is on the horizon. It’s currently available under compassionate grounds for patients who have exhausted other treatment options. Furthermore, regulatory bodies have begun to outline approval processes to aid in developing phage therapy.
Dr. Elkington is optimistic, envisioning that in 15 to 20 years, advancements could lead to phages being prescribed like antibiotics for specific infections.
If you’re curious about finding your own helpful virus, the Phage Collection Project will launch their new sampling kits during the Summer Science Exhibition, showcasing their innovative approach. “Antimicrobial resistance could touch us all,” noted Esme Brinsden from the Project. By getting involved, the public might just discover the next phage that can save lives.






















