Recently, there’s been quite a bit of back-and-forth regarding a medical research trial funded by the U.S. Many public health experts have classified the study as unethical and unnecessary, drawing comparisons to the notorious Tuskegee Experiment, whereas the Trump Administration has framed it as a rare chance to assess the potential negative effects of a vaccine.
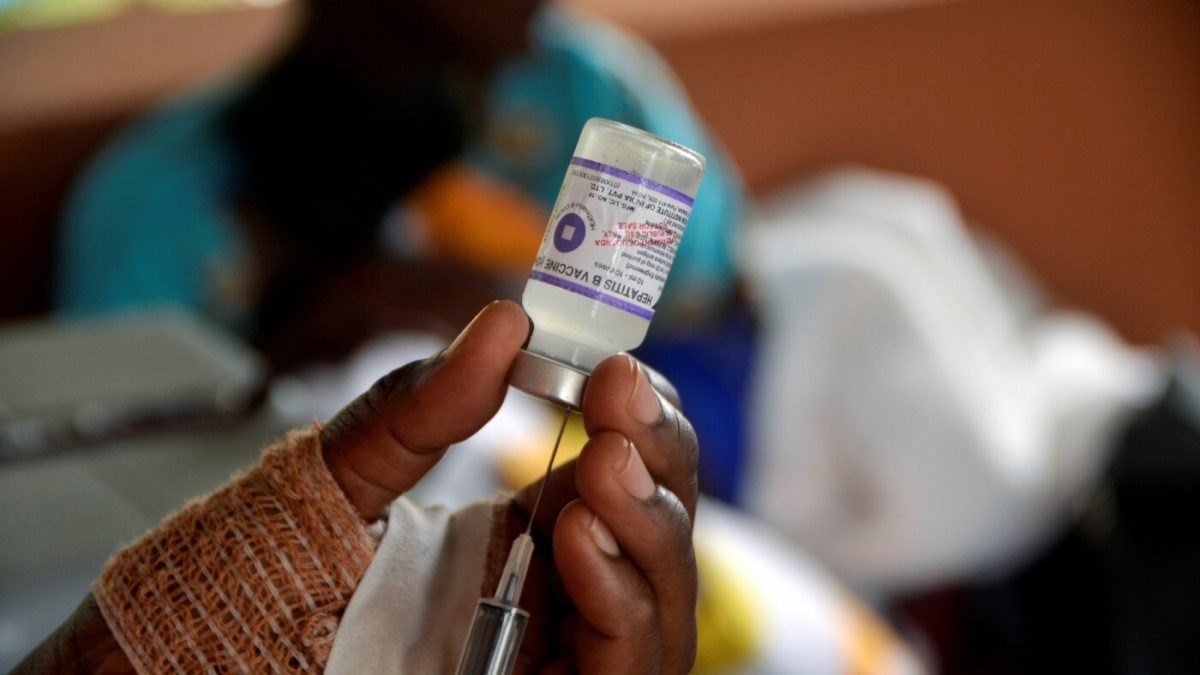
This trial, with partial funding of $1.6 million from the U.S. Centers for Disease Control and Prevention, aims to examine the general health impacts of administering the hepatitis B vaccine at birth in Guinea-Bissau, a country in West Africa.
For a relatively small study in a remote area, it’s received an unexpected level of scrutiny. There have been conflicting reports about whether the trial has started, been canceled, or is moving forward as intended.
This morning, at a press conference, Minister of Public Health Quinhin Nantote announced that the study is currently suspended while it undergoes review.
Nantote mentioned in Portuguese, through an interpreter, that they had either decided to suspend or possibly cancel the study. He noted that the local ethics committee that initially approved the study lacked the “required technical resources” and did not adequately coordinate with essential health authorities in the country. This situation arose partly due to the military coup in November, which resulted in many changes among high-ranking officials.
He also attributed the need for an ethics review to “misinformation, weak communication, and a lack of international dialogue,” all of which led to incorrect interpretations of the country’s health policies regarding immunizations.
The Africa CDC, the health agency of the African Union, has stated it will send a team to Guinea-Bissau to assist in evaluating the study, affirming that the final decision lies with local officials.
“It’s not up to a foreign nation to dictate whether this should take place. It’s about the country’s sovereignty,” Dr. Jean Kaseya, director-general of Africa CDC, stated.
Shortly after the press conference on Thursday, an anonymous official from the U.S. Department of Health and Human Services sent an email to NPR contradicting Nantote and Kaseya, insisting that the study has not been suspended.
This trial’s significance has captured attention for several reasons.
This study is happening as U.S. vaccine policy changes
For over 30 years, it’s been standard practice in the U.S. to administer the hepatitis B vaccine to all newborns, as recommended by the World Health Organization.
The vaccine protects infants from contracting hepatitis B from their mothers during birth. The virus targets the liver, potentially leading to cirrhosis, liver cancer, and death later in life. Currently, there’s no cure.
However, on December 5, CDC advisers voted to reassess U.S. policy.
These advisers, influenced by vaccine-skeptic and Health and Human Services Secretary Robert F. Kennedy Jr., expressed concerns that the vaccine might cause harm. However, many medical groups point to extensive studies showing its safety, noting that pediatric hepatitis B cases dropped significantly—by 99% since 1991, with only 10 reported cases in 2020 linked to birth exposure.
Not long after this vote, a document surfaced revealing plans for a CDC-funded study on the hepatitis B vaccine in Guinea-Bissau, where nearly 20% of adults are infected—far higher than the roughly 6% prevalence in sub-Saharan Africa.
The basics of the study
Guinea-Bissau currently doesn’t administer the hepatitis B vaccine to newborns, following WHO recommendations, but intends to start by 2028, according to Nantote.
Danish researchers plan to study 14,500 newborns during this “unique window” before birth vaccinations begin. Half will get the vaccine right after birth, while the other half will receive it later during routine vaccinations at 6, 10, and 14 weeks.
The goal isn’t to determine if the vaccine is effective, but rather to explore other health outcomes. They want to know if the vaccinated newborns face severe illnesses in the initial weeks, develop eczema by age two, or show neurological differences before reaching age five.
The ethics: unethical and unnecessary?
Once the study was announced, many medical professionals and ethicists called for its cancellation.
Dr. Paul Offit, director at the Vaccine Education Center at Philadelphia’s Children’s Hospital, described the study as “highly unethical and cruel,” likening it to the infamous Tuskegee Experiment, which denied treatment to African Americans in order to observe syphilis progression, leading to numerous deaths.
He argued that the cohort waiting six weeks for vaccination may encounter subpar healthcare and emphasized that these infants could be infected by their mothers before reaching that age.
Dr. Boghuma Titanji, an infectious disease physician, shared Offit’s concerns, highlighting that such studies could foster distrust towards medical intervention and vaccines within communities, a sentiment that can linger across generations.
In response, the researchers contended that no newborns in Guinea-Bissau would receive fewer vaccines due to the study, asserting that it could enhance vaccination coverage since trial teams will operate year-round.
Some ethicists raise the foundational question of whether the study addresses a significant query. Christine Grady, former chief of Bioethics at the NIH Clinical Center, noted that this might be the core issue, as the vaccine is already deemed safe and effective.
Researchers maintain that their findings will contribute to the wider scientific understanding of how vaccination impacts child health, implying that the trial could influence future vaccine guidelines globally.
The politics: Non-competitive, questionable researchers
Another contentious topic is the identity of the researchers and the nature of their selection.
The Danish team, led by Dr. Christine Stabell Benn from the University of Southern Denmark, has stirred controversy, with their studies facing scrutiny for alleged over-interpretation of findings.
In a recent journal evaluation, biostatisticians assessed Bandim Health research and found that the group frequently overstated conclusions that lacked solid statistical backing.
Stabell Benn’s team has challenged these findings and is seeking a retraction of the review.
Interestingly, Robert F. Kennedy Jr. supports Stabell Benn’s work, having cited a contentious study by her team while securing over a billion dollars in funding cuts from U.S. support for Gavi, a Vaccine Alliance.
Concerns have also arisen about the CDC granting $1.6 million for the project without an open competition, leading some to suspect that the Bandim Health Project was selected due to its connections to RFK Jr.’s vaccine-skeptic views.
Angela Rasmussen, a virologist, expressed concern over the implications of this connection, indicating that it poses risks to infants at a time when their health is paramount.
The U.S. HHS has defended the study, asserting it is moving forward as originally planned and stating it could represent a rare chance to evaluate the overall health effects of administering the hepatitis B vaccine at birth.