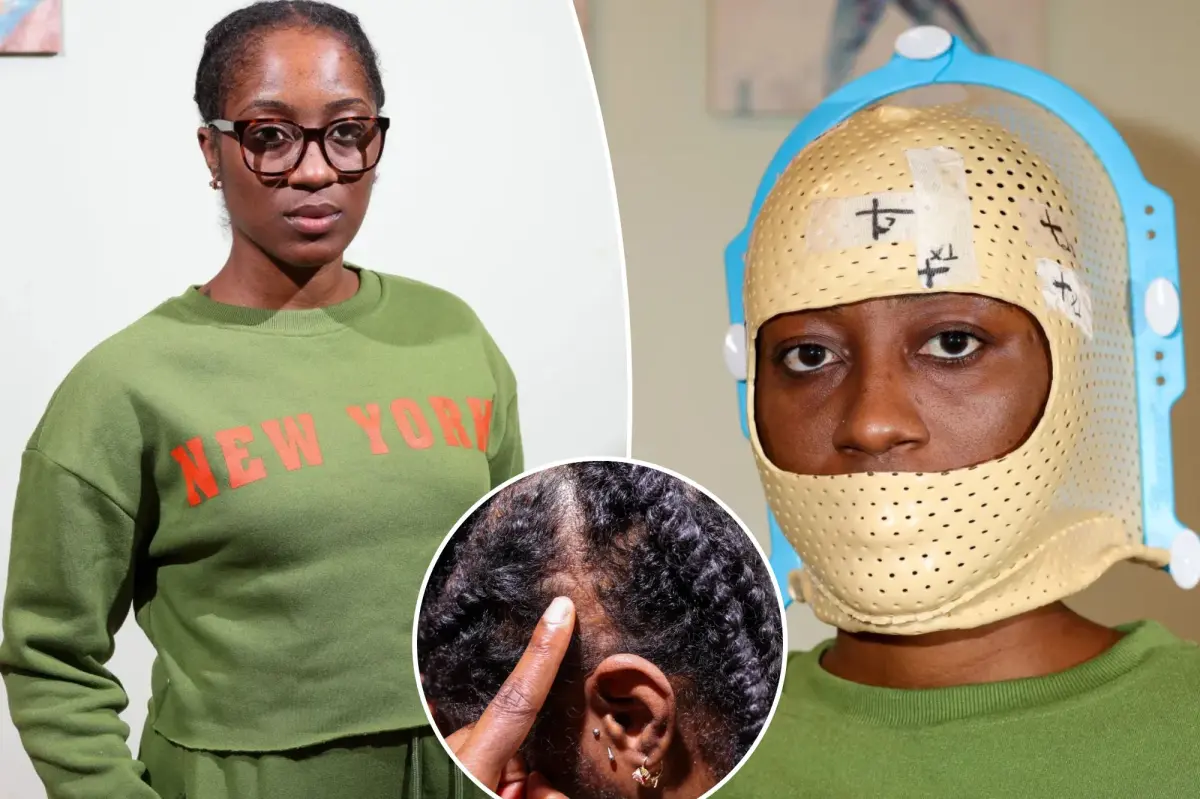
Elizabeth Frulisma breaks down in tears, her fingers tracing the scars on her head.
Her mother was taken aback to find out, at age 30, that she had a brain tumor about the size of a lime.
Though she survived a grueling 16-hour surgery followed by weeks of radiation, a part of the tumor still lingers, impacting her day-to-day life.
“Returning home after the surgery was like stepping into a foreign place,” shared Frulisma, who will turn 33 this week.
“Sometimes, when I speak, I find myself lost for words,” she reflected. “It’s challenging to recuperate from something like this and regain a normal life.”
Living in Centerreach, Frulisma suspects that her meningioma stemmed from nearly a decade of using Depo-Provera contraceptives.
She joins a group of 75 New Yorkers suing Pfizer, the manufacturer of Depo-Provera, claiming they failed to adequately inform users of the potential severe risk of developing brain tumors.
“In representing around 150 women and considering others, Elizabeth’s tumor stands out as the largest I’ve seen,” her attorney commented.
More than 1,700 individual lawsuits have surfaced in federal court, with plaintiffs asserting that Pfizer was aware— or should have been—of a significant risk for meningioma but chose to keep this information concealed to safeguard profits.
Pfizer responded, stating the allegations are baseless and they will defend themselves vigorously.
They added: “We continue to support the safety and efficacy of Depo-Provera, which is relied upon by millions of women globally and remains crucial for those seeking to manage their reproductive health.”
What is Depo-Provera?
Prioritizing her reproductive health, Frulisma gave birth to her first son at 18 in 2011.
Oral contraceptives didn’t suit her well, marking difficulty in consistent use.
Thus, her gynecologist recommended Depo-Provera, an injection given every three months, that many women have utilized.
The method contains progestin, a hormone that prevents pregnancy by stopping ovulation.
Research has shown that prolonged use of certain progestins, like medroxyprogesterone acetate found in Depo-Provera, raises the risk of developing intracranial meningiomas.
Frulisma’s complaint notes that the link between sex hormones and meningioma tumors traces back to the 1920s, with reports of progesterone receptors in tumors dating to the 1970s.
Several studies indicate that progesterone and its synthetic counterpart, progestin—particularly Depo-Provera—can lead to the development and growth of these tumors.
These slow-growing tumors arise from the meninges, the protective membranes for the brain and spinal cord. While mostly benign, they can press on nearby tissues or blood vessels.
Frulisma has no recollection of her doctor discussing any potential side effects of Depo-Provera, only noting that the pregnancy risk was minimal.
The potential side effects listed do include risks like reduced bone density and blood clots, but meningioma isn’t mentioned.
Pfizer states they urged the FDA to add a warning after a recent study indicated a possible link between Depo-Provera and meningioma, but their request was denied.
Inside Frulisma’s Emotional Battle
Frulisma began using Depo-Provera in 2014 at 21 and had no issues until early 2023.
“I was plagued by intense headaches and fatigue,” she recalled, with the pain reaching unbearable levels.
During one particularly troubling night, an elevated body temperature led her to the hospital, where the shocking discovery of a brain tumor was made.
“The news was staggering; we had never faced anything like this,” she said.
The ensuing months felt disorienting. She underwent a craniotomy at Stony Brook University Hospital in July 2023 but couldn’t have the entire tumor excised.
This meant she had to undergo radiation treatments five times a week for a month, totaling 27 sessions.
Each appointment lasted under two hours, and she wore a mask for radiation treatment, reminiscent of something from a sci-fi movie.
“The experience was mainly about lying there,” Frulisma explained.
Studying her surroundings, she recalled staff asking what music she wanted to help distract her, often thinking of vacations or favorite foods.
On the Road to Recovery
The path to recovery threw numerous challenges at Frulisma, from significant obstacles to small setbacks.
Being unable to care for her now-14-year-old son, she made the decision to have him stay with his father temporarily.
Witnessing the formation of her scar and the loss of hair was another painful aspect.
The fight isn’t over; she still consults with a radiologist annually to monitor any growth of the remaining tumor.
Frulisma disclosed that she continues to deal with vision issues, headaches, nausea, and dizziness.
Before this ordeal unfolded, she was a housing administrator aiding adults with intellectual disabilities.
“Once I got the diagnosis, the headaches made it impossible to work, and everything ceased,” she noted.
“I was in the midst of recertification as a nursing assistant and needed to train for mental health support, so all that was put on pause. Two years later, while things are reset, it’s been far from straightforward.”
Currently, she aspires to become a medical assistant with a focus on pediatrics.
Frulisma discontinued Depo-Provera in 2023, following her neurosurgeon’s advice.
She filed the lawsuit against Pfizer in March 2025, wanting to empower other women.
“Some women may hesitate to ask questions, but I didn’t,” she remarked. “It’s about taking charge of your health and advocating for yourself.”