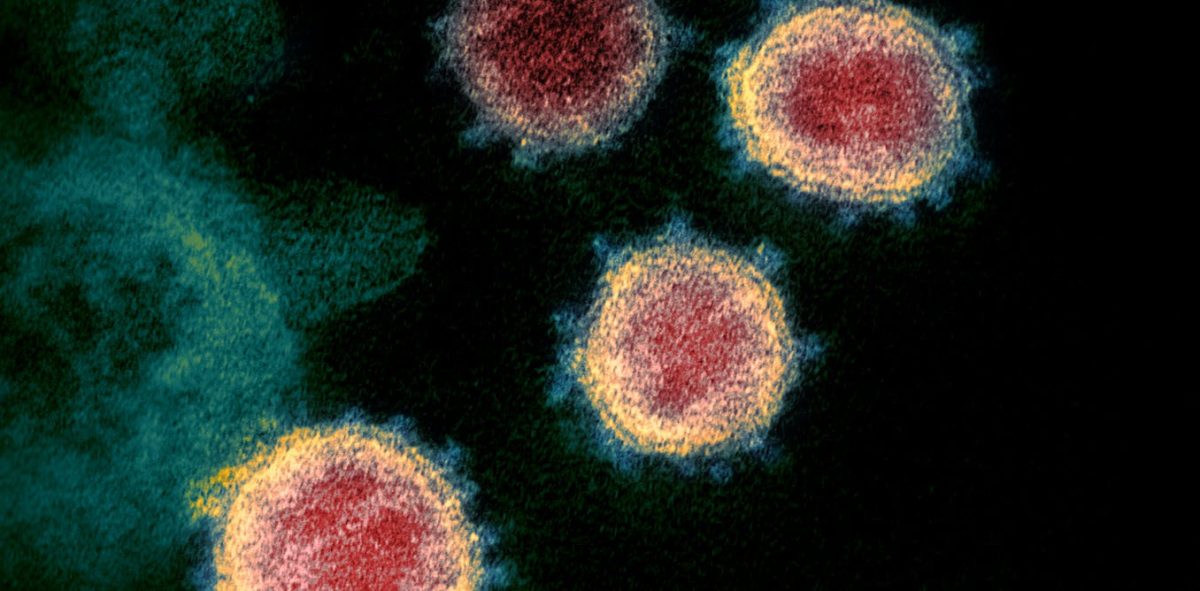
Many individuals who recover from infections like COVID-19, influenza, and glandular fever end up dealing with extended symptoms. Commonly reported issues include chronic fatigue, brain fog, exercise intolerance, dizziness, muscle or joint pain, and digestive troubles. Notably, these symptoms often intensify following physical activity, a condition referred to as post-exertional malaise.
Medically, these symptoms fall under the category of myalgic encephalomyelitis or chronic fatigue syndrome (ME/CFS). The World Health Organization categorizes this as a post-viral fatigue syndrome, recognizing it as a brain disorder alongside the United States Centers for Disease Control and Prevention.
While the occurrence of lingering illness post-infection is not a new phenomenon, the COVID-19 pandemic has magnified the issue globally. Almost half of those experiencing persistent post-COVID symptoms—collectively identified as long-COVID—meet the criteria for ME/CFS. Estimates suggest that since the pandemic began in 2020, over 400 million individuals have developed long-COVID.
To this point, there has been no widely accepted, testable explanation for the biological processes underpinning long-COVID and ME/CFS. Our research offers a fresh perspective that may contribute to understanding these conditions better.
Our research team focuses on blood and the cardiovascular system in the context of inflammatory and post-viral conditions, with particular attention to coagulation, inflammation, and endothelial cells. These cells form the inner layer of blood vessels and are crucial for functions like regulating blood clotting and managing inflammation.
In our latest review, we aim to clarify how ME/CFS and long-COVID develop and evolve, as well as how symptoms manifest throughout the body. By identifying and explaining the underlying mechanisms, we seek to enhance diagnostic and treatment tools for those affected by ME/CFS and long-COVID.
What is endothelial senescence?
In our review, our team suggests that certain viruses push endothelial cells into a dormant state referred to as cellular senescence, which resembles a “zombie” state. Senescent cells don’t divide anymore, but they continue to release molecules that disrupt and confuse the immune system, leading to increased blood clotting and constricted blood vessels.
By placing these “zombie” cells at the core of post-viral diseases, we link microclots, oxygen deficiency after exercise, brain fog, dizziness, digestive issues, and immune dysfunction into a cohesive, testable hypothesis.
From acute viral infection to ‘zombie’ vessels
Viruses such as SARS-CoV-2, the Epstein–Barr virus, HHV-6, influenza A, and enteroviruses can infect endothelial cells directly, compromising the cells that line blood vessels. Some of these viruses have been shown to induce endothelial senescence.
Research indicates that SARS-CoV-2 can trigger senescence in different cell types, including endothelial cells. Viral proteins from SARS-CoV-2 can hinder DNA repair pathways, steering host cells toward a senescent state, which in turn can make them more susceptible to further viral infections. This dynamic relationship sheds light on why various pathogens might lead to similar chronic conditions. Influenza A has also demonstrated the ability to induce a senescent state in endothelial cells.
What we think is happening
We believe that when blood vessel cells become “zombies,” they secrete substances that thicken the blood, heightening the risk of small clots. These clots can obstruct circulation, resulting in reduced oxygen supply to muscles and organs—a key reason for feelings of fatigue.
The problem becomes compounded during exercise. Instead of the blood vessels relaxing to allow for proper blood flow, they constrict further. This leads to oxygen deprivation in the muscles and a significant energy crash afterward. In the brain, the same dysfunctional cells lower blood flow, causing brain fog and dizziness.
In the digestive tract, these cells may weaken the lining, permitting bacteria to enter the bloodstream and incite further inflammation. Since blood vessels permeate the entire body, even isolated clusters of these senescent cells can generate the diverse symptoms seen in long-COVID and ME/CFS.
Immune exhaustion locks in the damage
Some immune system components, like natural killer cells, macrophages, and complement proteins, are tasked with eliminating senescent cells. However, individuals with long-COVID and ME/CFS often exhibit impaired function of these immune cells, leading to persistent senescent endothelial cells.
Senescent endothelial cells may also emit signals that inhibit immune response, enabling them to evade detection. This creates a cycle of vascular and immune dysfunction, allowing these “zombie cells” to thrive.
Under normal circumstances, a healthy immune system would clear these senescent cells. But the immune dysfunction seen in ME/CFS and long-COVID allows these “zombie cells” to persist and contribute to disease progression.
Where the research goes next
Currently, there’s a clinical trial in the US examining senescence in long-COVID. Our team is exploring innovative methods to detect aging signs in the cells lining blood vessels. Initially, we expose healthy endothelial cells in laboratory settings to blood from patients to see if it induces a senescent state.
Simultaneously, we are experimenting with non-invasive imaging and fluorescent probes that may eventually help identify these aging cells within the body. Occasionally, tissue biopsies might substantiate the findings from scans. Overall, these strategies aim to clarify how substances in the blood contribute to cellular aging, subsequently fostering disease.
Our ultimate goal is straightforward: identify these aging endothelial cells in actual patients. Detecting them can inform future clinical trials and pave the way for treatments specifically targeting senescent cells, potentially leading to improved vascular health and reduced disease burdens.