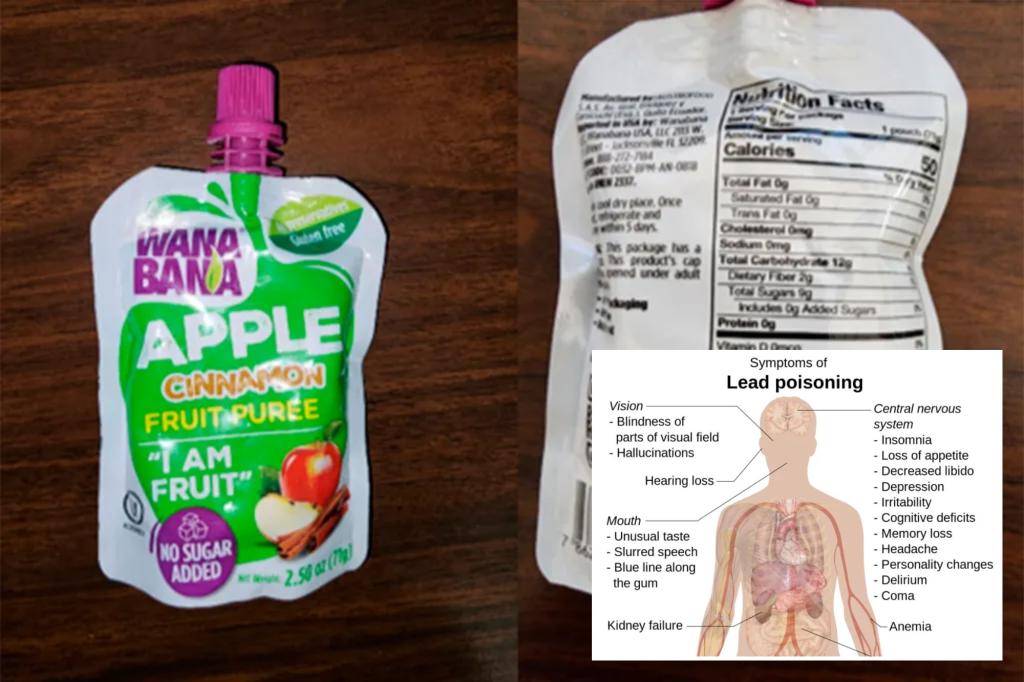
The parents of two North Carolina toddlers are suing Wannabana, claiming their children suffered lead poisoning after eating bags of the company’s allegedly contaminated fruit.
Nicole Peterson and Thomas Duong allege in a lawsuit filed Jan. 25 in Florida’s 11th Judicial Circuit Court in Miami-Dade County that their 1- and 3-year-old children have “potential developmental and They will require lifelong medical monitoring for health problems.” WanaBana has been incorporated.
“This is a nightmare that no parent should ever have to face. It is heartbreaking to know that our children will have to live with the effects of lead poisoning for the rest of their lives,” the couple said in a statement.
“This should serve as a wake-up call about the dangers lurking in everyday foods.”
Last October, WanaBana was hit with multiple class action lawsuits after announcing a recall of its Ecuadorian apple cinnamon fruit puree pouches.
Subsequent testing by the federal Food and Drug Administration found that the puree contained 2,000 times the maximum safe limit for lead. According to one of the class action lawsuit complaints.
Peterson and Duong, who are not part of the class action lawsuit, said they fed purees to their children for six months from February to August of last year, and that “the label on the product says it’s ‘kosher.’ According to the complaint, obtained by the Post, it is “additive-free” and “USDA organic.”
When the children were taken for annual exams, both blood tests revealed elevated lead levels, according to the complaint.
A follow-up visit three months later found that both infants’ blood lead levels had doubled, according to the complaint.
According to the Mayo ClinicFive micrograms per deciliter of lead in blood is a “potentially unsafe level for children,” causing developmental delays, learning difficulties, vomiting, and irritability, among other symptoms.
The couple’s children had radiation doses of 24.1 micrograms per deciliter and 20.8 micrograms per deciliter, respectively, according to the application.
According to the lawsuit, the concerned parents led an investigation into the lead levels at their child’s daycare center and also tested their own blood lead levels, which were found to be normal.
Peterson and Duong then implemented a so-called “elimination diet” (excluding food groups and slowly reintroducing them as part of a diagnostic procedure to identify the foods that cause the problem), which led to the “underage The only food the child consumes does not pose a problem.”consumed by [Peterson and Duong] According to the filing, these were defendant’s Wanabana fruit puree pouches. ”
The complaint says Peterson and Duong are seeking more than $50,000 in damages and also says the parents can take their case to a jury if the company doesn’t agree to a settlement.
Peterson and Duong said it is not yet known whether children will develop “symptoms of lead poisoning,” such as developmental or learning delays, as a result of elevated blood lead levels, so “ongoing treatment” is not recommended. argued that it would be necessary. Formative period.
The Post reached out to WanaBana for comment, but WanaBana has not issued any statement since the recall.
According to the parents’ attorneys at the law firm Motley Rice, Peterson and Duong were accused of using WanaBana’s products to cause elevated blood lead levels, which ultimately led to the Food and Drug Administration’s recall of the puree pouches. He was one of the first to report this.

The puree is sold across the United States and hit the shelves of Dollar Tree stores last December.
According to the class action lawsuit, the FDA has identified 87 complaints and reports of adverse events that may be related to the recalled products, with affected individuals ranging in age from infants to 53 years old.
An FDA spokesperson told the Post that the agency “does not comment on potential, pending, or ongoing litigation beyond what is publicly available.”