Potential for Microglia Replacement in Treating Brain Disorders
A potential treatment involving a fresh supply of immune cells known as microglia may someday address a variety of conditions, ranging from rare genetic disorders to more common issues like Alzheimer’s disease.
Recent studies have illuminated the prospects of a microglia replacement technique and have investigated ways to enhance its safety and effectiveness. “This approach is very promising,” remarks Pasqualina Colella, a gene and cell therapy researcher at Stanford University. “However, there’s a concern regarding the toxicity of the procedure.”
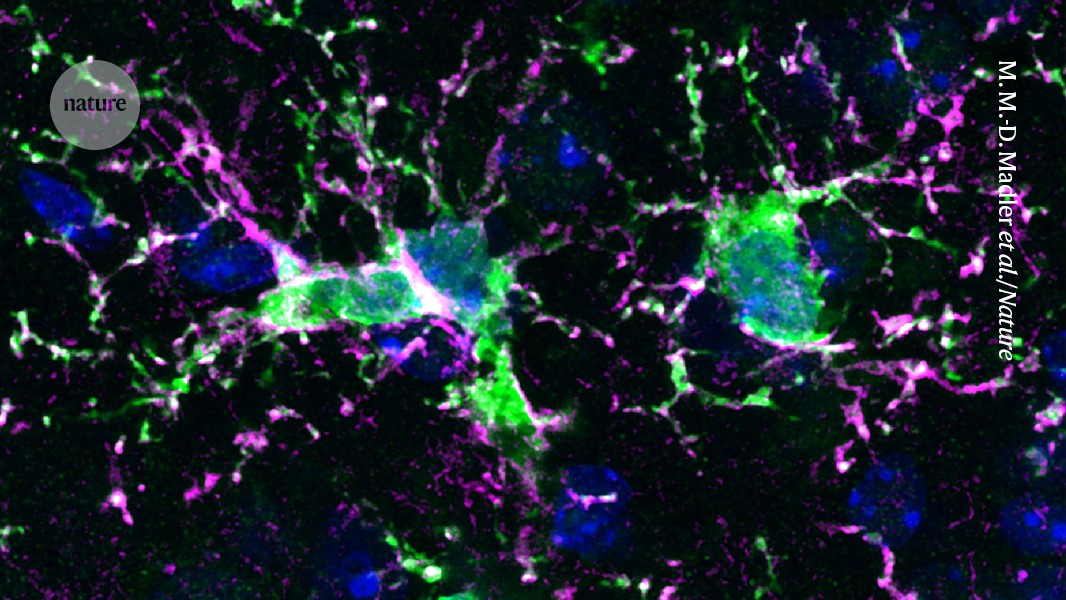
Microglia are essential immune cells that patrol the brain, absorbing foreign invaders, damaged cells, and toxins. They play a crucial role in shielding neurons during seizures and strokes and are involved in refining synaptic connections during typical brain development.
“Microglia perform several critical functions,” notes Chris Bennett, a psychiatrist specializing in microglia research at the Children’s Hospital of Philadelphia. “So, it makes sense that they are implicated in the development of numerous diseases.”
Diseases linked to microglial dysfunction include a range of rare conditions caused by genetic mutations, along with more prevalent disorders like Alzheimer’s and Parkinson’s disease, as well as aging-related cognitive decline, according to Bo Peng, a neuroscientist at Fudan University in Shanghai.
Challenges of Immune-Cell Replacement
This has prompted researchers to explore a compelling idea: could replacing dysfunctional microglia help treat brain ailments? Yet, this comes with its own set of hurdles. In typical cases, immune cells are replaced through bone-marrow transplants, which provide a new source of stem cells residing in the bone marrow that eventually give rise to various immune cells. In contrast, microglia are mainly found in the central nervous system and usually reproduce themselves rather than relying on stem cells for replenishment.
Currently, bone-marrow transplants are already in use for addressing specific rare diseases affecting microglia, such as X-linked adrenoleukodystrophy. While the treatment shows effectiveness, Marco Prinz, a neuropathologist at the University of Freiburg, cautions that outcomes can be inconsistent, with only a small percentage of the body’s natural microglia replaced.
In July, Peng’s team implemented bone-marrow transplants to replace defective microglia linked to a life-threatening brain illness known as CAMP (CSF1R-associated microgliopathy). Their success in both mice and a small study involving eight participants was promising: none experienced a decline in motor or cognitive abilities over two years, whereas those in a control group without treatment suffered deterioration.
One factor possibly contributing to the success of this trial is that individuals with CAMP tend to have low microglial production, potentially making space for the transplanted cells to flourish.
A Challenging Process
Creating an environment conducive to new microglia is pivotal yet concerning. To accommodate transplanted cells, physicians must first eliminate as many existing microglia as possible. This might require significant doses of chemotherapy or radiotherapy, which can heighten the risk for infection during the process and potentially increase long-term cancer risk. Hence, microglia replacement is currently deemed too toxic for use outside of severe, rapidly advancing conditions like CAMP, as noted by Colella.