Advancements in Lab-Grown Teeth Technology
It’s no secret that dental visits can be nerve-wracking for many. Replacing a tooth usually involves surgery, where a titanium screw is placed into the jawbone, followed by a long wait as it fuses into an artificial root before a crown is secured on top. Not exactly a pleasant experience, right?
However, researchers worldwide are exploring options to actually implant or even grow real biological teeth in a human jaw. It’s not something that’s happening overnight, but at King’s College London, Ana Angelova Volponi has been at the forefront of this research for nearly 20 years. In 2013, she contributed to a team that succeeded in growing a tooth using cells from both humans and mice.
This year, Volponi spearheaded a study that built upon those earlier findings and made significant strides in the material used to cultivate growing teeth in the lab. This new method closely mimics the natural environment of biological tooth growth in the mouth, marking a crucial step toward using human cells instead of mouse cells to generate teeth.
The idea of crafting lab-grown teeth isn’t entirely new; it traces back to the 1980s. According to Volponi, the initial breakthrough her team achieved over a decade ago was the first to utilize adult human gum cells—obtained by gently scraping the mouth—combined with “progenitor” tooth cells from a mouse embryo.
Volponi describes the process as being similar to a tripod, where two types of cells interact in a sort of dialogue to create a tooth, all within an appropriate environment.
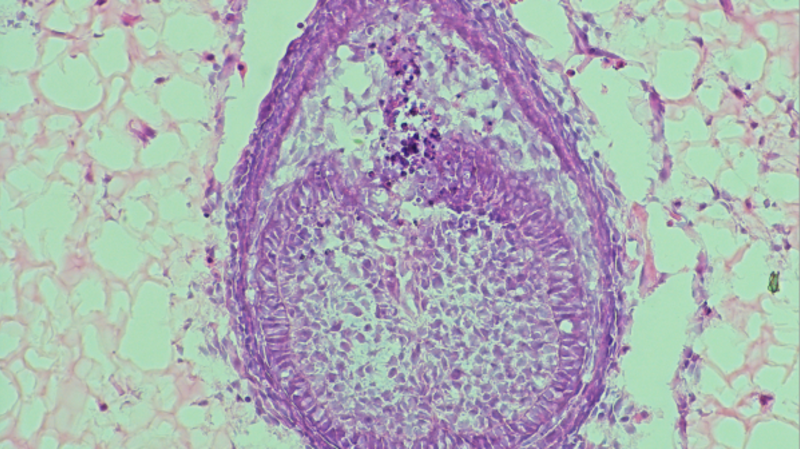
This environment, termed a “scaffold,” is vital for the formation of teeth in the lab. In her latest study, Volponi shifted from using a collagen-based scaffold to a hydrogel—a polymer with a high water content—explained doctoral student Xuechen Zhang, who co-authored the study. They first gather cells from mouse embryos, mix them to form a small pellet, and then inject this pellet into the hydrogel, allowing it to grow for about eight days. Interestingly, since the focus was on creating the environment, human cells weren’t necessary for this phase.
After those eight days, tooth-like structures emerge within the hydrogel, developed in collaboration with Imperial College London. Previously, in the 2013 study, these primitive tooth structures were placed into a mouse, where they continued to develop roots and enamel.
Of course, there are plenty of hurdles to clear before lab-grown teeth can become a viable option for patients. Yet, Volponi believes the new material enhances the interaction between the cells responsible for tooth creation.
Still, there are uncertainties about how to substitute embryonic mouse cells with adult human cells. Volponi sees two potential pathways for integrating lab-grown teeth into dental practice: either grow a tooth to a certain developmental stage before embedding it into the socket of a lost tooth, allowing it to fully mature into a biological tooth, or completely grow the tooth beforehand and then implant it surgically. She acknowledges it’s premature to determine which method may be more effective.
A biological replacement tooth crafted from a patient’s own cells could offer numerous benefits over traditional crowns or implants. For one, it would be more readily accepted by the body, avoiding inflammation or rejection, and would imitate the feeling and functionality of a natural tooth—unlike implants that often lack sensation and elasticity, merely fusing to the bone.
Vitor C. M. Neves, a senior clinical lecturer at the University of Sheffield’s School of Clinical Dentistry, emphasizes that Volponi has been a trailblazer in whole tooth regeneration, inspiring other researchers globally. He notes that her recent research addresses a key component for the production and potential commercialization of this technology—using matrices in whole tooth regeneration.
Neves adds that enhancing the lab environment for creating complete teeth is essential for clinical applications: “The more contributors there are in this field, the quicker we can realize the benefits for humanity.”
Others in the field are also pursuing different methods for tooth development.
For example, Katsu Takahashi’s team at Kitano Hospital in Osaka is working on an antibody-based treatment designed to encourage tooth growth in patients with conditions like anodontia, the absence of teeth. This treatment is currently in clinical trials and could be available by the decade’s end.
Meanwhile, in late 2024, a group led by Pamela Yelick at Tufts University succeeded in growing human-like teeth from a combination of human and pig cells in pigs, which are naturally able to regrow their teeth multiple times over their lifetimes. Their ultimate objective is to stimulate human cells to grow new teeth without involving any pig cells.
At the University of Washington, Hannele Ruohola-Baker and her team have successfully developed dental pulp stem cells from human stem cells taken from donated wisdom teeth. Their goal is to understand the molecular foundations of human tooth formation and replicate it in the lab. While Volponi’s work centers on creating tooth-like structures from existing tissues, Ruohola-Baker’s platform aims to generate the fundamental human tooth-forming cell types from scratch and guide them through proper development.
As for when these advancements might lead to tangible solutions, Ruohola-Baker is optimistic. “While clinical translation will take time, momentum in this field is accelerating, paving the way for biological tooth repair or replacement to become a real possibility within the next decade,” she says.