OAN Staff James Myers
11:10am – Tuesday, October 22, 2024
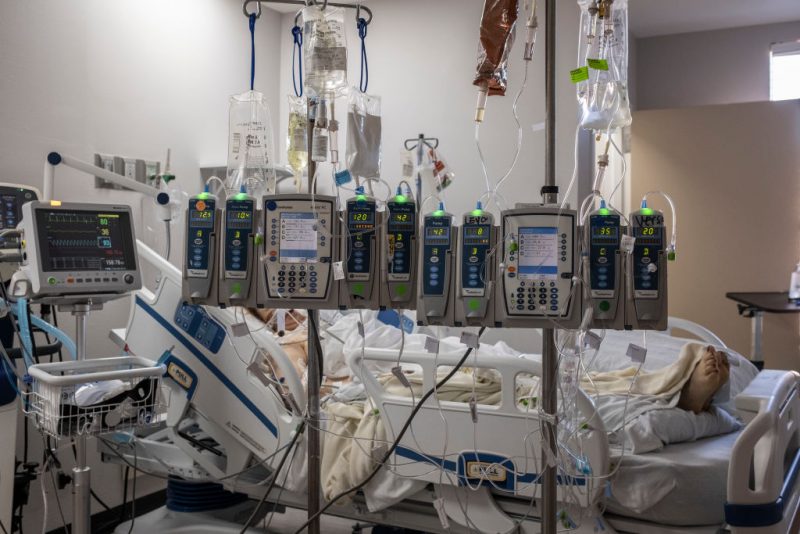
Multiple hospitals across the U.S. are reportedly dealing with significant issues related to intravenous fluid shortages after flooding from Hurricane Helen forced medical fluid manufacturers to halt production.
advertisement
“A factory involved in making intravenous fluids for one of the country's largest manufacturers has been flooded by Hurricane Helen,” the BBC reported.
Baxter International, a leading supplier of intravenous fluids to hospitals, announced that its facility in Marion, North Carolina, will remain closed until further notice.
“Baxter's North Cove facility was affected by flooding and production is currently suspended as we assess the extent of the impact,” the company said in a statement on its website.
IV fluids are used to deliver water containing medications or electrolytes directly into a patient's bloodstream. It is also important during surgery to keep you hydrated if you are unable to eat or drink for some reason. Baxter also makes specialty fluids such as peritoneal dialysis fluid, which helps kidney failure patients filter waste from the blood, and irrigation fluids used during procedures to clean or flush wounds.
“Remediation efforts are already underway and we are devoting human and financial resources to help restart production and ensure patients and healthcare workers have the products they need.” said Jose (Joe) E. Almeida, President and Chief Executive Officer. Baxter said in a statement.
Additionally, Brigham General, Mass., is among the health systems that do not have a regular supply of IV fluids. Chief Readiness and Continuity Officer Dr. Paul Bidder said in a press conference Friday that the hospital network will receive about 40% of what it normally receives from Baxter.
Bidder said the supply constraints are “one of the biggest shortages” the hospital network has ever dealt with.
Meanwhile, other IV fluid makers say they are ramping up production to make up for the shortage. B. Brown said its manufacturing sites were unaffected by Hurricane Helen and that it is “focusing on critical IV fluids and increasing production at its pharmaceutical manufacturing sites in Irvine, Calif., and Daytona Beach, Fla.” We are taking immediate action.”
Additionally, the company has placed its products on “protective allocation” and is currently encouraging healthcare providers who administer IV fluids to practice protections, such as using alternative hydration methods.
In 2017, Hurricane Maria halted production and hospitals faced a shortage of IV fluids.
“Having experienced similar challenges in the aftermath of Hurricane Maria in 2017, we remain mindful of how we manage the supply of these medicines to minimize the impact on patients. “Hospital operations continue as normal and patient care is unaffected,” Dr. Bidder, head of the Massachusetts Brigham General Command, said in a statement.
Stay informed. Receive breaking news directly to your inbox for free. Subscribe here. https://www.oann.com/alerts
Advertisement below
Please share this post!