Most Trump Voters Believe FDA Should Safeguard Americans from Dangerous Chinese Medications

Trump Voters Concerned About Unsafe Prescription Drugs A recent poll by Fabrizio Ward, associated with President Trump’s voter base, shows that a significant majority of Trump supporters are very worried about the potential dangers posed by unsafe foreign medications entering the U.S. via online pharmacies and counterfeit products. The findings highlight widespread bipartisan agreement on […]
Understanding kratom: FDA focuses on harmful 7-OH items in nationwide enforcement

U.S. health agencies are addressing the dangers posed by substances that are widely accessible across the country. One such substance is 7-hydroxymitraginine, commonly referred to as 7-OH. This plant-derived compound is often used for pain relief but has also been linked to potential abuse and various adverse drug reactions. Health officials have categorized this substance […]
FDA advises classifying concentrated Kratom as an illegal drug

The Trump administration has proposed that 7-OH, a compound derived from the Kratom plant, be classified as an illegal substance, as announced by Food and Drug Director Marty McCurry on Tuesday. This compound, a by-product of Kratom, is often marketed as a dietary supplement that can enhance energy levels or alleviate pain. FDA officials indicated […]
The FDA needs to take action against hazardous counterfeit weight-loss medications.
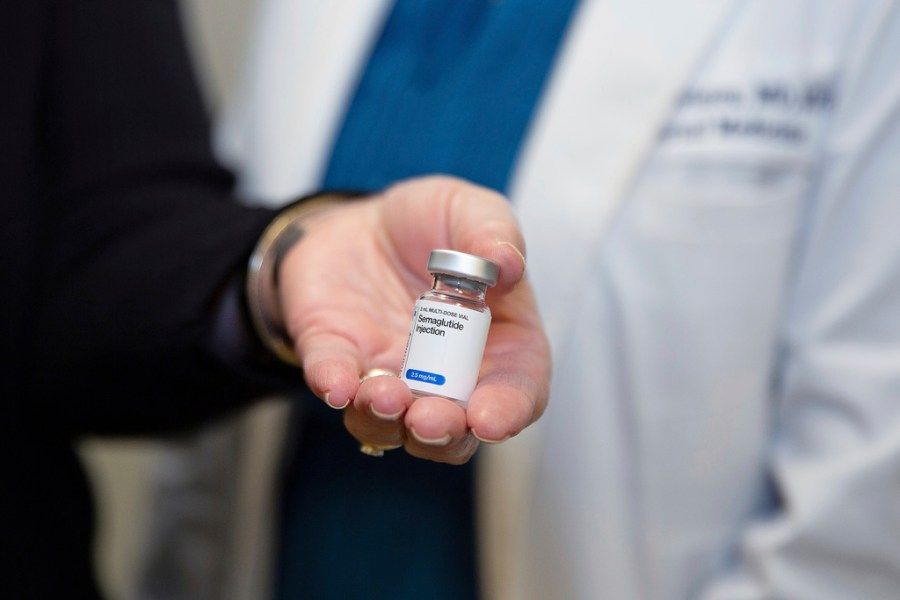
Hope in the Fight Against Obesity For the first time, there’s a glimmer of hope in tackling obesity. Recent data from the National Center for Health Statistics at the CDC indicates that adult obesity rates in the US might finally stabilize after years of annual increases since at least 2011. Obesity, long understood to be […]
FDA Chief To Hear Concerns About Forced Use Of Abortion Pill Readily Accessible Online

FDA Commissioner Discusses Mifepristone Review In a recent conversation with Politico’s Dasha Burn, Commissioner Marty McCurry shared his insights on Mifepristone, the abortion medication. He noted that he observes data indicating that patients sometimes take the drug outside the supervision of their healthcare providers. During the interview, which is set to air completely on Sunday’s […]
FDA Chief Warns Fluoride in Water and Supplements Might Harm Internal Organs

FDA Commissioner Raises Concerns Over Fluoride in Drinking Water Marty McCurry, the Commissioner of the Food and Drug Administration (FDA), has expressed serious concerns about the chemicals used in fluoride for drinking water. He described their potential effects on children’s organs as akin to “carpet bombing.” During a speech for the American Research Fund, McCurry […]
FDA changes its decision on Juul

FDA Reverses Juul’s Ban The Food and Drug Administration (FDA) has allowed Juul Labs to resume selling its e-cigarettes, nearly three years after the company faced a ban on its products. It’s a significant shift that many are watching closely. The FDA announced that Juul now has permission to market its original e-cigarette device along […]
Gardenia Blue Color Gets FDA Approval in Victory for MAHA

FDA Approves New Natural Food Color The U.S. Food and Drug Administration (FDA) announced on Monday that it has approved a new natural food color, making it the fourth such addition under the Trump administration’s American Health (MAHA) initiative to phase out artificial food dyes in the U.S. The newly approved color, Gardenia (Jenipin) blue, […]
Michelle Pfeiffer criticizes ‘very concerning’ food technology approved by the FDA that was once supported by Bill Gates

Michelle Pfeiffer Raises Concerns Over APEEL Michelle Pfeiffer is expressing her worries about APEEL, a food spray brand. On Thursday, she took to her Instagram story to voice her concerns after the FDA approved its use on USDA-certified organic produce. “APEEL, which consists of edible plant-based coatings meant to extend the shelf life of fresh […]
FDA Chief: “Our priority is food”

As part of the “Make America Healthy” initiative, Dr. Marty Makary, the Commissioner of the Food and Drug Administration (FDA), has announced a significant goal: eliminating artificial food dyes by the end of 2026. During a recent interview with News Nation, McCurry, who was appointed by President Trump as the 27th FDA chief, reflected on […]









