Breakthrough in Gene Therapy for Rare Condition
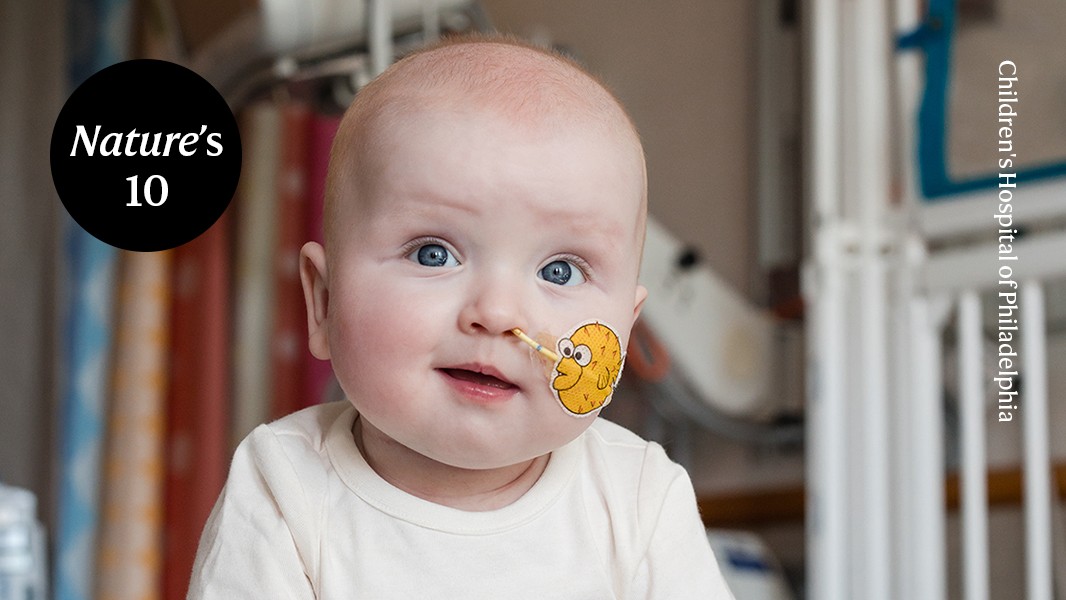
KJ Muldoon, initially referred to as Patient Eta by his medical team, has become the face of a significant medical breakthrough. Just a few months after his birth, KJ’s image, along with his bright, cheerful smile, was featured in news outlets globally as the first individual to undergo a personalized CRISPR-based genome-editing therapy.
After KJ’s birth in August 2024, his doctors observed that he was excessively sleepy and not feeding well. A series of tests revealed that he has a very rare condition known as carbamoyl-phosphate synthetase 1 (CPS1) deficiency, which hampers protein processing in the body.
When proteins break down, they produce ammonia, a toxic substance typically handled by enzymes in the liver and expelled through urine. In CPS1 deficiency, one of these crucial enzymes falters, leading to ammonia build-up in the bloodstream that can harm the brain. The primary treatment is a liver transplant, but tragically, about half of the infants with CPS1 deficiency do not survive past early infancy.
Dr. Rebecca Ahrens-Nicklas, a pediatrician at the Children’s Hospital of Philadelphia, speculated that correcting the faulty enzyme in KJ’s liver might be an alternative. Teaming up with Kiran Musunuru, a cardiologist from the Perelman School of Medicine at the University of Pennsylvania, they devised an ambitious plan to treat children with rare genetic disorders through gene-editing therapies customized to their unique DNA sequences, with KJ positioned to be the first to benefit.
Unlike previous gene-editing therapies that aimed at broader populations—like Casgevy, the first approved CRISPR therapy for widespread blood disorders—KJ’s treatment was specifically designed just for him.
The research team employed a variant of CRISPR known as base editing to address the problematic genetic mutation—literally correcting one faulty DNA letter out of three billion. Such a highly personalized treatment had never been created so rapidly, and KJ’s situation was urgent; his body could succumb to ammonia toxicity in a matter of months.
It required a vast collaboration among researchers from various sectors to bring this plan to fruition. While KJ’s playful nature delighted hospital staff, Musunuru and his team consciously avoided learning personal details about him, such as his name. “We had critical decision points where objectivity was essential based on the data,” Musunuru explained.
Manufacturing teams worked tirelessly to produce the necessary gene-editing components for KJ’s treatment. “We initially projected that it would take 18 months,” shared Sandy Ottensmann from Integrated DNA Technologies in Iowa. “We managed to complete it in six.”
On February 25, KJ received the first of three infusions. His ability to tolerate protein has improved, though he will still need ongoing medication and monitoring to keep his ammonia levels stable.