It took eight years for cardiology organizations to update their guidelines, aligning them with new evidence on lipid panels that now include lipoprotein(a) — Lp(a) and Apolipoprotein B — ApoB. It’s a bit alarming, really, to think of how many high-risk individuals for atherosclerosis and cardiovascular events were overlooked during this time due to outdated guidelines. Almost two years ago, in this space, I discussed “The Under-Appreciation of CHIP,” emphasizing the importance of this assay, which delivers significant risk insights regarding cardiovascular disease, cancer, blood clots, and more. If you’re curious about this, I’d recommend checking that out since I won’t delve into much backstory here. Instead, I’m going to summarize some important new data on CHIP, share my findings, and advocate for action.
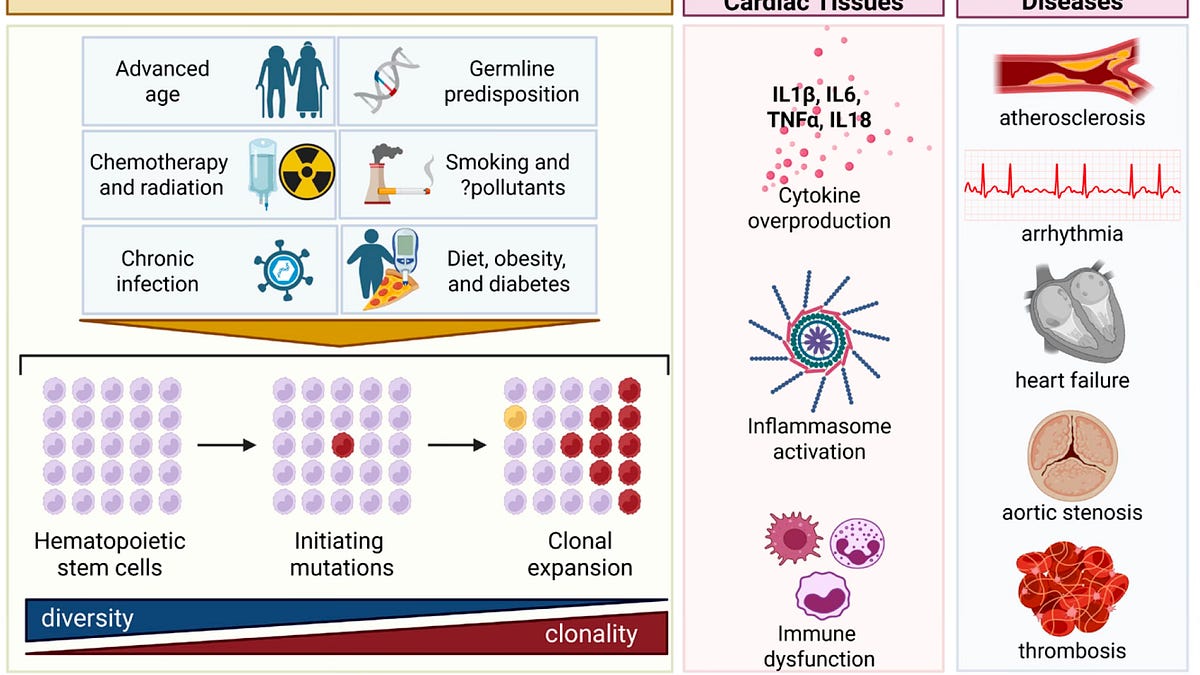
CHIP stands for clonal hematopoiesis of indeterminate potential. It involves mutations in blood stem cells, leading to clonal expansion, defined by a cutoff of 2% or greater of blood cells. Aging increases the prevalence of CHIP: around 2% in those aged 50-69, 10% from 70-79, 20% in the 80-89 range, and over 39% for those aged 90 and above. The most common mutations seen are in DNMT3A (about 48%), TET2 (around 22%), and ASXL1 (11%), along with others at roughly 19%. The primary mutations are found in epigenetic genes. The presence of CHIP is increasingly linked to several diseases, especially cardiovascular issues, cancers, and inflammatory conditions, with risk rising alongside CHIP frequency. Just last month, the American Heart Association recognized CHIP as a potential contributor to cardiovascular disease.
Aside from age, CHIP risk factors include inflammation and immune dysfunction, influenced by aging — a phenomenon known as inflammaging and immunosenescence. Although only about 0.5% to 1.0% of individuals with CHIP above 10% develop blood cancer annually, underlying immune dysfunction and a constant pro-inflammatory state can still be detected. In patients with advanced solid cancers, over 25% exhibit CHIP, significantly higher than in age-matched healthy controls. As noted in my previous discussions, the cardiovascular disease hazard ratio associated with CHIP is 1.8, higher than that of well-known risk factors like smoking and hypertension, which sit at 1.4. A new risk score for CHIP is now available, allowing better assessment of risk levels.
I’ll categorize these findings into disease and treatment-related discoveries, though this is just a snapshot of recent research emphasizing the significance of CHIP.
To add context, some findings suggest TET2 mutations can enhance responses to checkpoint therapy in lung and colorectal cancer, revealing that circulating CHIPs can affect tumor microenvironments by promoting T-cell responses. However, in another study involving TET2 mutations and aortic aneurysms, a link was found between pro-inflammatory effects and accelerated aneurysm growth. A major advancement in understanding CHIP’s resilience was the identification of a gene variant on chromosome 17 that reduces various CHIP subtypes and blood cancers, linked to down-regulation of the RNA binding protein MSI-2. This could pave the way for interventions to mitigate CHIP and its negative health consequences.
On to treatment findings: low-dose colchicine was found to consistently reduce CHIP in a randomized placebo-controlled trial. This anti-inflammatory medication has been previously examined in atherosclerosis models and TET2 mutation patient cohorts. Further, therapies like IL-1β–neutralizing antibodies have shown efficacy in reducing events for participants with TET2 mutations. Other potential treatments include metformin, Vitamin C, and inhibitors of interleukins like IL-6, among others. An ongoing clinical trial is also assessing the NLRP3 inflammasome inhibitor selnoflast. Interestingly, even GLP-1 medications, known for their anti-inflammatory properties, are being explored as potential anti-CHIP options.
I personally underwent a CHIP test at no cost, courtesy of a company called Lucence. My results showed a CH level of just 0.3%, well under the 2% threshold. The only detected driver mutations were TET2 variants, each at low VAF. Compared to the general population, my results suggested a lower risk for major outcomes, and given that over 10% of my age group has CHIP, I feel quite fortunate. If I had tested positive for TET2 CHIP, I might have considered starting colchicine.
We’re discovering more about CHIP and its associations with numerous diseases, including cardiovascular and thromboembolic conditions. Different mutations present varying risks, and even the same TET2 mutation can sometimes result in both harmful and beneficial effects. Research, including animal models, supports the idea that CHIP isn’t just a passive observer but may actually drive adverse outcomes.
So, why don’t we routinely test for CHIP in older adults? One reason could be the lack of effective therapies, but that argument becomes weaker when considering treatments like low-dose colchicine. Also, accurately assessing CHIP can be tricky; while whole genome sequencing offers a way, the best method is using targeted panels with deep sequencing. Lucence charged $950 for my test, which is frankly too high. Given recent decreases in sequencing costs, developing an affordable CHIP assay should urgently be prioritized alongside trials for drugs that show promise.
There’s also the concern that CHIP testing could cause anxiety in patients, similar to what cancer survivors experience. However, identifying risks and implementing preventive measures often outweighs potential downsides. It’s often better to fend off disease than to treat it afterward.
With this backdrop in mind, it’s significant that a couple of academic institutions have established dedicated CHIP Clinics, like those at the University of Chicago and the Cleveland Clinic. After my last post, I came to learn about a hybrid cardio-oncology CHIP clinic at Yale Medicine, but I can’t help but wonder — why aren’t there more? With all this compelling evidence, why aren’t we making CHIP assessments widely available and affordable for older individuals? Blood markers for inflammation can be misleading, and I’m inclined to change the term to Clonal Hematopoiesis of Important Potential—getting rid of that “indeterminate” label. After all, it seems quite informative to me. Will it really take another eight years for guidelines from relevant professional societies on diagnosing and treating CHIP?