Thymic Health and Immuno-Oncology: A Study Overview
This study highlights the significance of thymic health in the context of immuno-oncology. We analyzed data from 3,476 cancer patients treated with immune checkpoint inhibitors (ICIs) and discovered that better thymic health was linked to improved patient outcomes. It was particularly notable for lung cancer and melanoma, which are frequently treated with immunotherapy. Interestingly, similar associations were found in breast and renal cancer, suggesting that thymic health is a universal prognostic indicator, regardless of tumor type. Further analysis of the TRACERx cohort indicated that measuring thymic health through imaging can serve as a proxy for its functionality and the overall competence of the immune system. These results point out that a person’s immune health might significantly influence immunotherapy success, emphasizing the need to integrate host-specific factors as biomarkers instead of relying solely on tumor-centric ones.
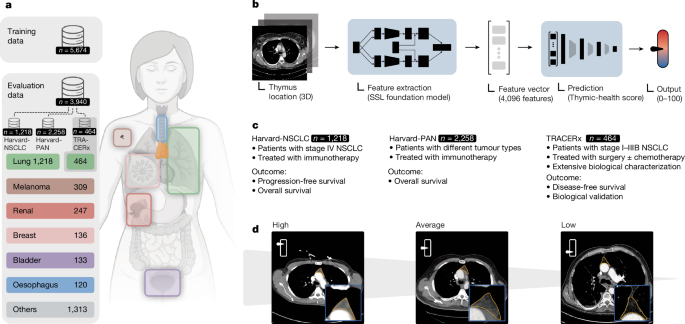
Although immunotherapy is becoming more prevalent, not all patients respond positively. Improving biomarkers is essential to better estimate individual prognoses. Currently, most approaches focus predominantly on cancer-specific biological markers, despite the crucial role T cells play in the effectiveness of ICIs. We define thymic health as an important prognostic biomarker based on the radiographic features of the thymus, which is essential for T cell maturation and the generation of a diverse T cell repertoire. These features are vital for recognizing foreign antigens and responding to immunotherapy.
Our findings suggest that thymic health can enhance prognostication in immuno-oncology using standard thoracic CT scans, without requiring additional imaging or causing extra stress to patients. More research is needed to define specific thresholds for this biomarker and to clarify its predictive values. Nevertheless, the evidence indicates that thymic health could be a key marker in immuno-oncology, possibly functioning independently or in tandem with other established measures, such as PD-L1 and tumor mutational burden (TMB). While PD-L1 remains a solid predictor of response, our research indicates that thymic health might add value beyond PD-L1 by reflecting the host’s immune competence.
We identify different cancer patients who may benefit from immunotherapy. The benefit of high thymic health appears to vary across cancer types, with some showing improved outcomes at both high and average levels of thymic health. In contrast, others only showed better results with high levels. This variability indicates that thymic health operates on a gradient influenced by tumor type, immune responsiveness, and specific treatment protocols. Similar patterns have been observed with TMB, where certain cancers respond differently to immunotherapy depending on the TMB predictive value. Our findings suggest that evaluating thymic health can aid in pinpointing patients at risk for poor outcomes, highlighting the importance of preventing non-beneficial immunotherapy to enhance quality of life and reduce unnecessary risks.
The poorer clinical outcomes observed in patients with low thymic health are likely linked to reduced naive T cell production. Analysis in the TRACERx cohort confirmed that patients with higher thymic health displayed significantly greater levels of sjTRECs, indicators of thymic activity. Additionally, a correlation between higher thymic health and greater T cell diversity supports previous findings about the link between thymus status and T cell diversity. Thus, the condition of the adaptive immune system is pivotal for treatment outcomes, emphasizing the role of thymic characteristics in immune capacity.
It’s noteworthy that thymic health was found to be prognostic in advanced non-small-cell lung cancer (NSCLC) patients receiving immunotherapy, even among those previously treated with chemotherapy. The effects were most pronounced in first-line therapy settings, with a consistent trend toward the importance of thymic health prior to chemotherapy. Given that chemotherapy can impair thymic function, maintaining thymic health may influence long-term outcomes from systemic cancer treatments. The ongoing prognostic value in pretreated patients highlights thymic health as a reliable marker of immune competence.
Our results may have broader implications for personalized cancer therapy, particularly regarding the balance of immune cell types that shifts with age. Evidence from animal models suggests that specific depletion of certain immune cell types can rejuvenate the immune system, and similar results may be achievable in humans through targeted therapies aimed at improving thymic function. Nonetheless, rigorous preclinical studies are essential before any large-scale implementation.
Previous research indicated that thymic involution, or decline in thymic health, correlates with increased disease risk in adults. Attempts to visually assess thymic involution through CT scans yielded basic associative results that align with our findings, showing variations in thymic health based on factors like age, sex, and smoking. In contrast to previous notions that most adults experience thymic degeneration, our data showed that higher and average thymic health was related to better outcomes in a significant portion of the examined population.
Our study utilized data from various cohorts, implementing analyses that reveal a strong association between thymic health and treatment outcomes in diverse cancers treated with immunotherapy. This suggests a need for assessing thymic health as a factor in patient stratification for immunotherapy. Moreover, this has implications beyond cancer treatment to current practices in cardiothoracic surgery and radiation oncology. Often, the thymus isn’t regarded as essential and can be inadvertently removed or subjected to high-dose irradiation. This research implies potential benefits from retaining the thymus, yet additional studies are crucial to ascertain in which clinical contexts this is relevant.
One limitation of the study is the predominantly white demographic of the sample, necessitating further validation across diverse populations. For the thymic-health model to be implemented clinically, it must show generalizability across varied scanners and settings. Although the current approach has demonstrated robustness and high stability, further work in appropriately powered cohorts is required to determine if thymic health can act as an independent biomarker for immunotherapy response. This reinforces the notion that thymic health is a vital component of tumor-agnostic immunotherapy efficacy.
In conclusion, this study sheds light on the previously unrecognized importance of the thymus in immuno-oncology. Our findings, supported by other studies, indicate that immune system status is unique to each individual and varies by various factors, further influencing response to immunotherapy. Most existing biomarkers focus specifically on cancer types and are largely tumor-derived; in contrast, assessing thymic health offers a more holistic, host-specific alternative through non-invasive imaging techniques. This insight could spur further research on the relevance of the thymus in maintaining health against cancer and beyond, paving the way for a personalized medicine approach that integrates overall patient health with relevant tumor biomarkers for enhanced outcomes.