When his young son was diagnosed with a rare, fatal disease, a Canadian father was dejected to learn there was no treatment or cure, so he decided to create one himself.
Terry Pirovolakis, an IT director from Toronto, Ontario, Canada, welcomed his third son in December 2017. He told Fox News Digital that it was a “normal, healthy birth,” but less than six months later, he and his wife, Georgia Pirovolakis, noticed that baby Michael wasn't holding his head up.
“It didn't seem like he was hitting his target,” Pilovolakis said.
Mother desperate to save clinical trial that could cure daughter: 'The cure is in my fridge'
After months of doctor visits, physical therapy and genetic testing (what Pirovolakis describes as an “18-month diagnostic journey”), neurologists diagnosed baby Michael with spastic paraplegia 50 (SPG50), a neurological disorder that affects fewer than 100 people worldwide.
“The doctors told us to just go home and love our son. They also said he would be paralyzed from the waist down by the age of 10 and a quadriplegic by the age of 20,” Pirovolakis said.
When Michael Pirovolakis, pictured, was diagnosed with a rare, fatal disease as a toddler, his father, Terry Pirovolakis, was devastated to learn there was no treatment or cure. That's when he decided to create his own treatment. (Terry Pilovolakis)
“He can't walk or talk and doctors have told him he will need assistance for the rest of his life.”
What is SPG50?
Spastic Paraplegia 50 (SPG50) Child DevelopmentAccording to the National Organization for Rare Disorders, it can gradually lead to cognitive impairment, muscle weakness, speech problems and paralysis.
Most people with the disease die before they reach their 20s.
“Children with SPG50 may experience early developmental delays, muscle weakness and spasticity, but they continue to work hard and adapt,” Dr. Eve Elizabeth Penny, an epidemiologist with the Texas Department of State Health Services and a medical contributor to DrugWatch, told Fox News Digital.
What is Angelman Syndrome? Colin Farrell's son lives with the rare condition
“Over time, these symptoms can worsen and patients may have difficulty walking and carrying out daily activities,” added Penney, who was not involved in Michael Pirovolakis' treatment.
“The prognosis varies from person to person, but in general it is a progressive disease and symptoms can become more severe over time,” she said.

Georgia Pirovolakis (left) is shown with her two sons, including baby Michael, who was diagnosed with SPG50. (Terry Pilovolakis)
In the absence of treatment, most families will seek physical therapy, occupational therapy, speech therapy, Drugs that can help Penny said it could help control spasticity and seizures.
“Managing SPG50 requires a comprehensive, multidisciplinary approach to address the range of symptoms and challenges,” she added.
A Father's Mission
Currently, there are no U.S. Food and Drug Administration (FDA)-approved treatments for SPG50.
After being shocked by the diagnosis, Pirovolakis immediately began research, focusing on finding a gene therapy that could help his son.
“I was told that by the age of 10 I would be paralyzed from the waist down and by the age of 20 I would be a quadriplegic.”
A month after her baby's diagnosis, Pirovolakis flew to Washington, DC, for a conference on gene therapy, where she met with several experts, and also visited the National Institutes of Health in Sheffield and Cambridge University in the UK, where scientists were studying the disease.
“We then took all of our savings, refinanced our house and paid a team at the University of Texas Southwestern Medical Center to run a proof-of-concept study to begin gene therapy for Michael,” Pirovolakis said.

Terry Pirovolakis, pictured with his family, used his life's savings to develop a gene therapy for his youngest son (center), who has SPG50. (Terry Pilovolakis)
After tests proved the gene therapy was effective in halting the disease's progression in mice and human cells, Pirovolakis worked with a small Spanish pharmaceutical company to manufacture the drug.
On December 30, 2021, Health Canada approved Michael Pirovolakis' gene therapy to move forward.
Stiff-Person Syndrome Patient Explains What It's Like Living with a Rare Disease
“On March 24, 2022, my son became the first person to receive gene therapy at SickKids Hospital in Toronto,” Pilovolakis said.
Although this procedure, in which cerebrospinal fluid is injected via a spinal tap, carries risks, its potential benefit is lifesaving.
“I couldn't let them die.”
After Michael Pirovolakis received one dose, he had three more doses remaining.
“We decided we had to help other kids,” Pilovolakis said.
“When I heard that no one was going to do anything, I knew I couldn't let them die.”

Pilovolakis' two older children (pictured below left with their brother Michael) do not have the disease. (Terry Pilovolakis)
Pirovolakis began a phase 2 trial in the United States two years ago and has treated three children.
One of them was 6-month-old Jack Lockard, the youngest person to receive the treatment.
“Since then, Jack has thrived,” the boy's mother, Rebecca Lockard, told Fox News Digital.
The Girl Who Couldn't Smile: How a Rare Disease Became One Young Woman's 'Great Gift'
“He's sitting up on his own, banging around toys, drinking from a straw cup and crawling with all his might.”
She adds: “Doctors and therapists are on the same page: treatment works!”
The other children Participated in the test Lockard said he has experienced similar results.

The Lockard family, pictured here, are struggling to raise money for medical treatment for their daughter Naomi (right), who has SPG50. (Rebecca Lockard)
“We saw that all of them had their disease progression halted and their cognitive abilities improved.”
As Fox News Digital previously reported, there are other children in need of treatment, including Lockard's first child, 3-year-old Naomi, who also has SPG50 and cannot receive the treatment because clinical trial funding has dried up.
“Time is of the essence.”
It costs about $1 million to make enough medicine for one child, and another $300,000 to treat a patient in a U.S. hospital, Pirovolakis said.
Pirovolakis approached pharmaceutical companies, but they all declined to manufacture the drug.
“We want to make sure this trial goes ahead and children have access to treatment.”
“Investors are not going to fund treatments for diseases that are not profitable,” he said. “That's the dilemma we find ourselves in.”
While Pilovolakis and his team have been active in securing grants and investors, that’s largely the responsibility of parents. Raise funds On to the next stage of clinical trials.

So far, Lockard has raised more than $90,000 through a GoFundMe account (Naomi vs Jack Fight SPG50) for her daughter's medical expenses, but this is only a fraction of what is needed. (Rebecca Lockard)
So far, Lockard has raised more than $90,000 through a GoFundMe account (Naomi vs Jack Fight SPG50) for her daughter's medical expenses, but this is only a fraction of what is needed.
Developing a treatment for SPG50 will be difficult and expensive, Penney said, “mainly because it's a sporadic disease.”
“Pharmaceutical companies often prioritise diseases that affect larger populations where they have a better chance of recouping their R&D costs,” the doctor told Fox News Digital.
'Breakthrough' gene therapy restores hearing for deaf children: 'It's like a miracle'
“The market is much smaller. Rare diseases It becomes financially difficult for companies to invest in developing treatments, as SPG50 has done.”
To focus on the work, Pilovolakis quit his job and founded a nonprofit in California that now has five employees and 20 consultants.
The company, named Elpida Therapeutics after the Greek word for “hope,” plans to begin a Phase 3 trial of SPG50 at the NIH in November.

Terry Pilovolakis, second from left, is pictured with team members from the nonprofit Elpida Therapeutics, which has partnered with the Columbus Children's Foundation (Fundación Columbus in Spain) and CureSPG50 to help children suffering from the disease. (Pirovolakis)
Without the support of Major pharmaceutical companiesBut there are no funds to provide treatment to children who need it.
Eight doses of the SPG50 drug were manufactured in Spain and airlifted to the United States.
“The treatment is here, it's literally in the fridge and it's ready to go,” Lockard said. “The doctors are ready to go. We just don't have the funding to make it happen.”
For more health related articles, health
Pilovolakis said there are currently four families in the U.S. trying to raise the necessary funds.
“Time is of the essence,” he said. “We want the trial to move forward and for the kids to get treatment.”
The End Goal
Looking ahead to the Phase 3 clinical trial at NIH, Pirovolakis' goal is to treat eight children with SPG50.
“If we can prove that it works for all eight kids, and if we can prove that it works to the FDA, the drug will be approved and all kids will have access to it,” he said.

Pictured here is Michael Pirovolakis walking with the help of a walker. Spastic Paraplegia 50 (SPG50) is a neurological disorder that affects a child's development, gradually causing cognitive impairment, muscle weakness, speech problems and paralysis. (Terry Pilovolakis)
Ideally, once the drug is approved (which Pirovolakis estimates could take three to five years), SPG50 will be used in hospitals to treat patients with Newborn Screening Program And all children with the disease will have access to treatment.
Elpida Therapeutics has partnered with the Columbus Children's Foundation (Fundación Columbus in Spain) and CureSPG50 to work to help children suffering from this disease.
Click here to get the FOX News app
“Our partnership with Elpida is based on our unwavering commitment to leaving no child behind,” CCF co-founder Sheila Mikhail said in a statement to Fox News Digital.
“As global organizations, Columbus Children's Foundation and The Columbus Foundation believe every child deserves the chance to build a healthy future. Together, we are making groundbreaking advances in treating ultra-rare genetic diseases to ensure no child faces these challenges alone.”
“The biggest challenge in providing treatment to children with rare diseases is often a lack of funding and vision.”
Pilovolakis said she receives several calls each week from families around the world pleading for help to save their children.
“Unfortunately, the biggest challenge in providing treatment to children with rare diseases is often a lack of funding and vision,” he told Fox News Digital.
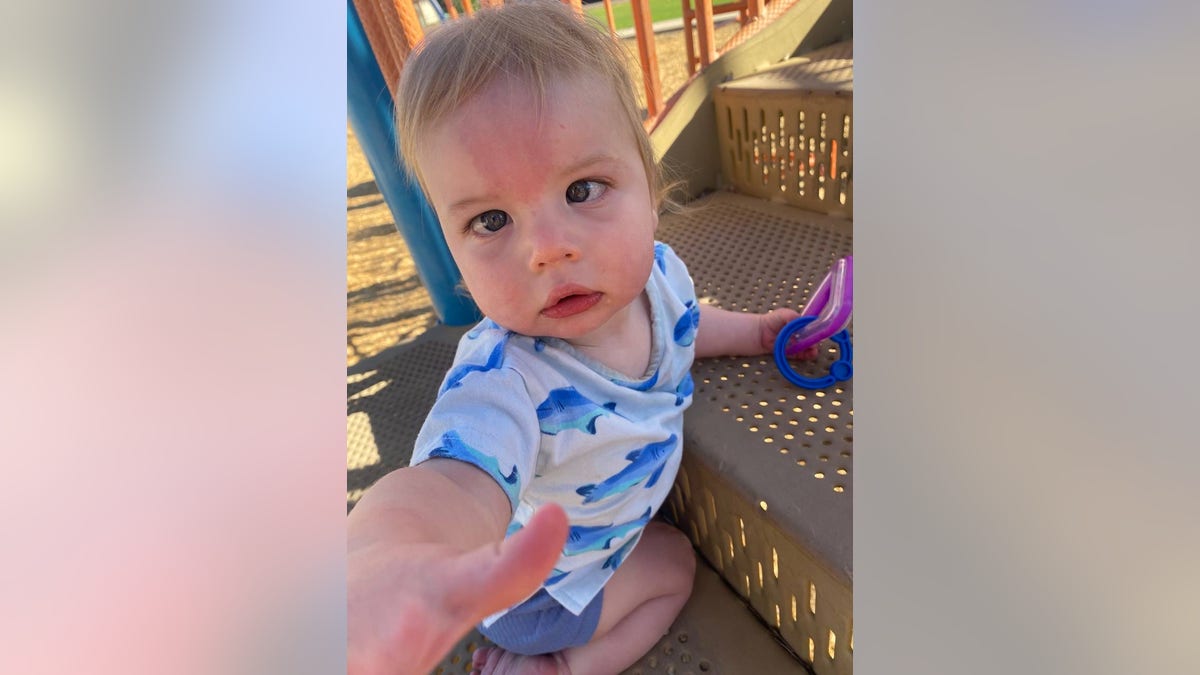
After Jack Lockard, pictured, underwent gene therapy at six months old, his family immediately noticed improvements in his cognitive and physical abilities. (Rebecca Lockard)
“The technology is already there to treat children. The hope is that someone with enormous wealth and, more importantly, vision and influence will come in,” he said.
“Their support will not only impact a few diseases and a few children, but it will give hope to thousands of rare diseases and millions of children, this generation and the next.”
Click here to sign up for our Health Newsletter
Currently, 40 million Americans are living with a rare disease, meaning that one in 10 suffers from a treatable rare disease.
“There's a good chance that someone you know or love has a rare disease,” Pirovolakis added.






















