Three Individuals Sue Vaccine Manufacturers Over Severe Reactions
Three individuals are filing lawsuits against vaccine manufacturers alleging serious injuries sustained during Covid vaccine trials. The plaintiffs, who discussed their situation with the Daily Mail, claim the companies dismissed their injuries and failed to cover related medical expenses, despite having contracts in place indicating they would.
In 2020, over 200,000 people volunteered to participate in clinical trials to evaluate the safety and effectiveness of vaccines for Covid. While many participants only experienced mild side effects like fatigue and headaches, a few assert that they endured lasting, life-altering injuries. They allege that the manufacturers neglected to acknowledge their serious side effects and did not provide medical assistance or compensation.
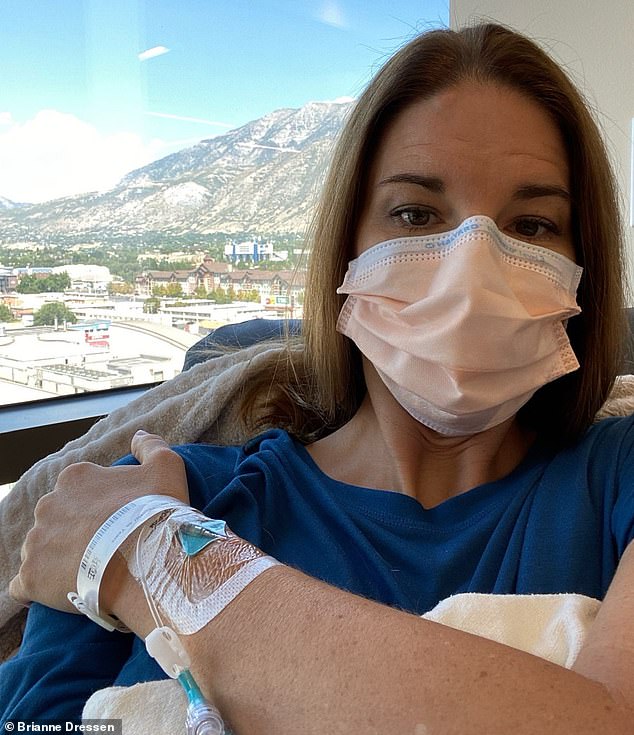
Among those suing is Brianne Dressen, a 39-year-old mother and former preschool owner from Utah. After receiving a single dose of the AstraZeneca vaccine in November 2020, she developed alarming symptoms. Within an hour, she experienced tingling that spread throughout her body and described it as a “horrific electrical pulsating sensation” that persisted around the clock. Dressen stated that her condition deteriorated to the point where she couldn’t walk, and was hypersensitive to light and sound for four months — even casual interactions with her children became overwhelmingly painful.
Despite her ongoing struggle with more than 20 symptoms, including autoimmune nerve damage and fluctuating heart rates, AstraZeneca allegedly failed to meet contractual obligations for her care. Her husband, Dr. Brian Dressen, a chemist, pursued answers for her plight and found some help from a specialist at the National Institute of Neurological Disorders and Stroke. Ultimately, she was diagnosed with post-vaccine neuropathy during a trial set up to investigate potential vaccine injuries.
Over the following year, Dressen attended numerous medical appointments and faced mounting bills. While she sought financial support from AstraZeneca as per her agreement, she only received a minimal offer linked to relinquishing their liability for future claims. This prompted her to pursue a lawsuit against the company.
AstraZeneca has yet to respond publicly but previously filed to dismiss the case, a motion that was denied, leading to an appeal.
In the US, the PREP Act was enacted to grant immunity to vaccine manufacturers during public health emergencies, which complicates efforts for those injured to seek damages. Claims for injuries associated with emergency use vaccines must be filed within 12 months through a compensation program established for cases like these, although many claims have been rejected.
For instance, Kleiton Luis de Oliveira Souza, age 24, participated in AstraZeneca’s trials and developed epilepsy after his second dose. He reported severe symptoms and even required hospitalization, with doctors concluding his injuries were vaccine-related. Like Dressen, Souza found AstraZeneca unresponsive to his injury claims.
In another trial, Augusto Roux, then 36, experienced severe health complications after receiving the Pfizer vaccine in Argentina. He endured extended periods of high fever and significant heart issues, but claims that his reactions were downplayed by trial staff. Despite reassurances from Pfizer that they take adverse effects seriously, Roux’s experience reflects concerns over the handling of such incidents during trials.
While there is broad agreement among experts that vaccine-related injuries are rare, the small percentage of affected individuals remains crucial to consider. They stress the importance of rigorous follow-up and acknowledgment of any adverse effects experienced by trial participants.
Calls for further investigation into both the vaccine trials and their aftermath appear to be growing more urgent as these cases unfold. It’s a complex issue, and while the benefits of vaccinations in preventing Covid are acknowledged, ensuring transparency and care for those who experience adverse effects is equally significant.