Two Gut Bacteria Might Be Linked to Multiple Sclerosis—But It’s Early Days
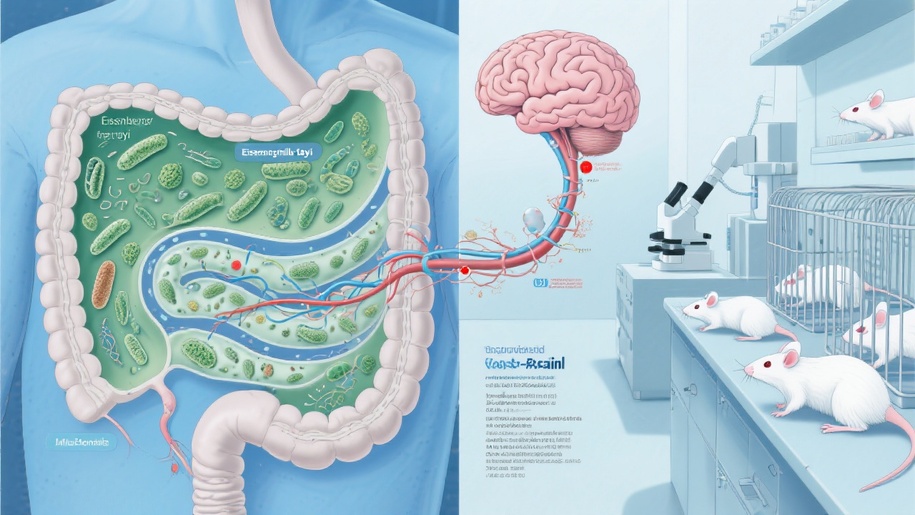
Nearly a million people in the U.S. deal with multiple sclerosis (MS), a condition where the immune system mistakenly attacks the brain and spinal cord. Researchers have long suspected that gut health could be involved in triggering this autoimmune response. A recent twin study has identified two specific bacterial suspects: Eisenbergiella tayi and Lachnoclostridium.
This study, published in PNAS, focused on 81 pairs of identical twins, with only one twin diagnosed with MS. This setup is particularly smart because it controls for genetic factors while revealing different health outcomes. The research team found that the twins with MS had higher levels of these two bacteria in their small intestines.
But it gets more intriguing from here.
Connecting Gut Health to Neurodegenerative Issues: Mouse Experiments
The researchers took the gut bacteria from the twins and transplanted them into mice that were specially bred to develop MS-like symptoms. The results were pretty striking: mice that received bacteria from MS twins developed paralysis within about three months, while those that got bacteria from healthy twins remained active.
Interestingly, female mice experienced worse effects—this aligns with the real-world observation that women are three times more likely to develop MS than men.
E. tayi and Lachnoclostridium are typically harmless members of the Lachnospiraceae family, which help digest fiber. However, what’s setting these two apart is still unclear, though they can consume mucus when fiber is scarce—this might lead to a weaker intestinal barrier and expose immune cells to inflammatory signals.
A Broader Perspective: MS Statistics
Multiple sclerosis affects about 2.9 million people globally, with nearly a million living in the U.S. It usually manifests during pivotal life stages, with an average diagnosis age of 32 when people are often establishing their careers and families.
The disease is unpredictable. About 85% of those diagnosed initially experience relapsing-remitting MS, where symptoms come and go. Without treatment, around half of these individuals will progress to secondary progressive MS within ten years, leading to cumulative disability.
Current therapies can slow the disease’s advancement but don’t cure or reverse the damage. This is why identifying specific bacterial triggers is significant—it could provide a targeted approach rather than attempting to overhaul the entire gut biome.
Reality Check: Is This a Major Breakthrough?
This isn’t the first time gut bacteria have been linked to MS. Previous studies identified different bacterial patterns with similar implications. The field is crowded with promising connections that have not necessarily borne fruit.
There are a few concerns to take into account:
The mouse model limitation: What happens in mice doesn’t always translate to humans. Numerous treatments that show success with rodents have failed in human trials, mostly due to differences in immune systems, gut environments, and the sterile conditions in which lab mice live.
Sample size: While 81 twin pairs might seem significant, it’s still fairly small for drawing wide-ranging conclusions about such a complex disease.
Causation versus correlation: Even after the mouse experiments, we’re not entirely sure if these bacteria cause MS or if they merely thrive in the inflammatory environment that the disease creates.
The need for replication: Other research teams must validate these findings in various populations. Science often includes exciting results that, unfortunately, can’t be duplicated.
Looking Ahead
If these findings hold true, the therapeutic possibilities could be compelling. Targeted antibiotics, tailored bacteriophages, or specially designed probiotics might eliminate the problematic bacteria while preserving beneficial ones.
Early trials of high-fiber diets have shown some slight improvements in MS patients, possibly by keeping mucus-consuming bacteria busy with safer food choices.
However, any therapy aimed at the gut will have to navigate the delicate ecosystem of beneficial bacteria responsible for vitamin production and immune regulation. Broad-spectrum antibiotics, for instance, can sometimes exacerbate autoimmune diseases, emphasizing the need for balance.
The Bottom Line
This twin study exemplifies structured scientific research. The researchers accounted for genetics, employed functional testing in mice, and focused on specific bacterial species instead of broadly indicating “microbiome imbalances.”
However, it remains just one study with a small sample size. History has shown us that MS research often leads to dead ends. Remember when some believed that a virus caused MS? Or when it was thought to be purely genetic?
It seems reasonable to predict that this research will lead to follow-up studies—some will affirm the findings, while others may provide contradicting evidence. That’s how science generally unfolds—messy, gradual, and frequently disappointing.
For the millions living with MS, this research offers a glimmer of hope for new treatment avenues. But it’s important to stress that it’s not a definitive breakthrough yet—it’s merely a promising lead requiring years of further investigation to validate.
The connection between the gut and the brain in autoimmune conditions is an important area of study. As to whether these two bacteria are the main players or just minor roles in a much bigger story, that’s still to be determined.