CDC Advisory Committee Shifts Covid-19 Vaccine Recommendations
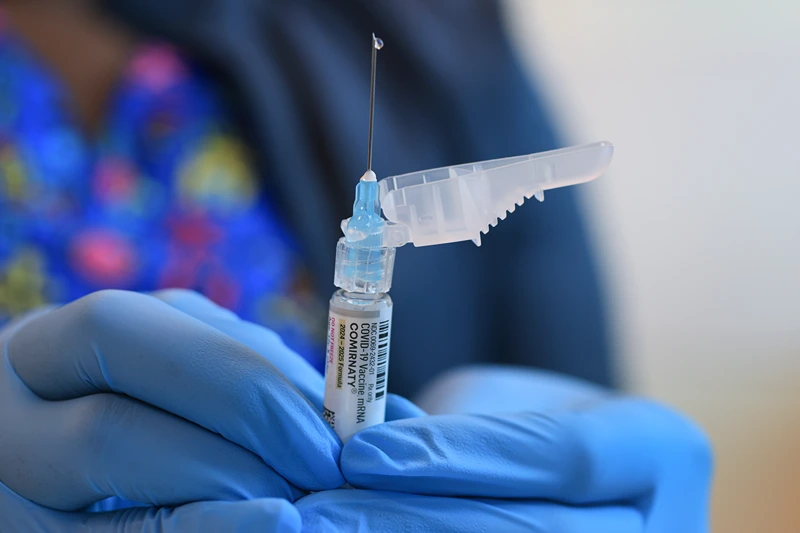
The Centers for Disease Control and Prevention’s Vaccine Advisory Committee has made a significant change, deciding unanimously to stop recommending the Covid-19 vaccine as a routine option. Instead, they emphasized that vaccination should be based on personal decisions tailored to individual circumstances.
Previously, nearly all Americans who wanted the Covid-19 vaccine received it, following the CDC’s earlier guidelines.
The new recommendations will prioritize individuals over 65 years old and those in higher risk groups, especially in light of recent limitations on the use of the Pfizer, Moderna, and Novavax vaccines.
Now, individuals aged 6 months to 64 are encouraged to make personal vaccination decisions based on the data available.
One committee member noted the shift, saying, “In previous Covid seasons, getting vaccinated was as easy as stopping at the pharmacy or doctor’s office. It was a regular recommendation.”
Another member remarked that this vote signifies that the Covid-19 vaccine is “no longer routinely recommended,” indicating a transition to “shared clinical decisions” and a more focused set of recommendations.
In a separate decision, the committee rejected a requirement for prescriptions to obtain the vaccine, with the vote tied at 6-6. The tie was broken by ACIP chair Martin Kulldorff, who voted against the requirement.
Furthermore, the CDC voted to ensure clearer communication regarding the risks associated with the Covid-19 vaccine, which can be shared with patients at the time of vaccination.
The discussion on the Covid-19 vaccine was led by ACIP member Retsef Levi, an MIT professor. Levi had been chosen to head the CDC’s Covid-19 working group after expressing skepticism about the vaccines’ rapid rollout, suggesting they may not have proven effective and could contribute to higher mortality rates.