Hope Amid ALS Challenges
Jeff Vierstra experienced a heartbreaking family history; his mother and two sisters succumbed to complications from ALS, or Amyotrophic Lateral Sclerosis. At 41, he’s now looking to an experimental treatment that might just be a groundbreaking attempt to prevent this neurodegenerative disease from affecting him.
When his mother passed away, Vierstra was merely two years old. Tragically, all her siblings also lost their lives to ALS in their thirties and forties.
Despite his interests in skiing—like bravely heading down an iceberg in Chile—or skydiving in British Columbia, it’s his family’s medical history that truly unnerves him. Along with his sisters, Erin and Leigh, Vierstra discovered that they all carry a mutation of a gene called “FUS,” vital for nerve cell functionality. This mutation significantly increases their risk of developing ALS.
“Having that kind of cloud hanging over you is mentally and emotionally really challenging,” Vierstra remarked.
Research indicates that around 10 to 15% of ALS patients have a genetic variant of the disease, with about two-thirds of these being familial, which means multiple family generations are affected. Dr. Neil Shneider, a neurologist at Columbia University specializing in genetic ALS, noted this concerning trend.
According to the Centers for Disease Control and Prevention, approximately 35,000 individuals in the U.S. are currently living with ALS, commonly known as Lou Gehrig’s disease. The illness progressively deteriorates motor neurons, the nerve cells responsible for muscle movement. Over time, patients typically lose their capacity to speak, walk, and eventually breathe.
While there isn’t a cure as of now, many researchers globally are searching for effective treatments. Among these efforts, an experimental method is being developed at the Eleanor and Lou Gehrig ALS Center at Columbia University for a rare genetic variant of the disease.
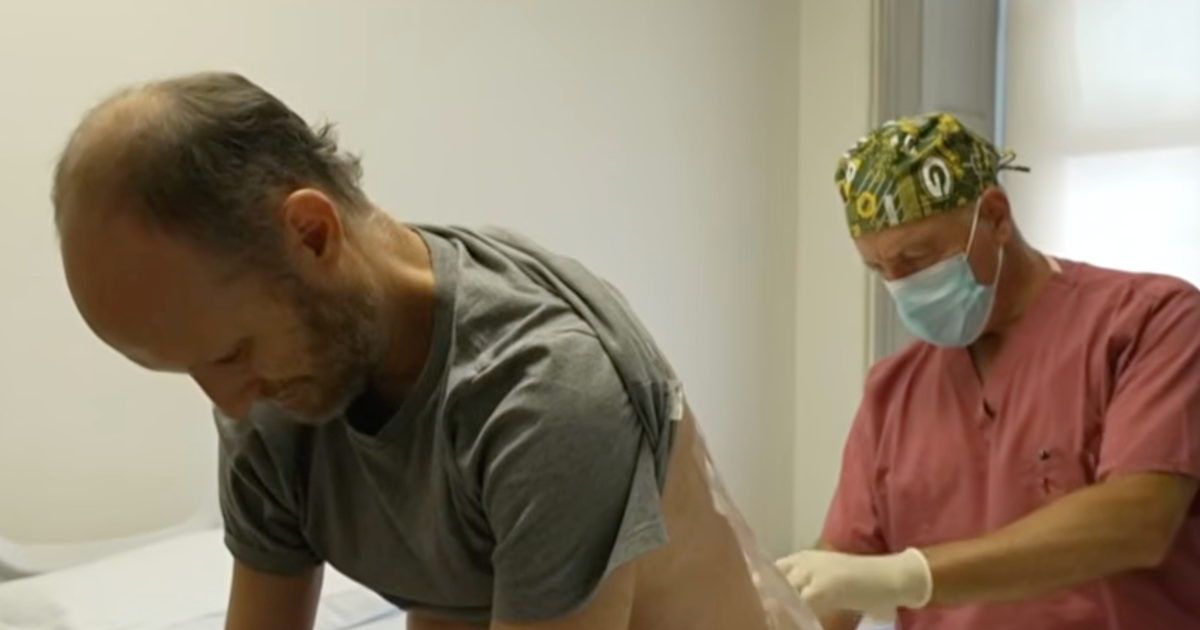
Both of Vierstra’s sisters began showing ALS symptoms and participated in a clinical trial at Columbia for an experimental treatment aimed at addressing the mutated gene. Dr. Shneider, who oversees the trial, assessed Vierstra and detected irregularities in his EMG (electromyography) tests, which monitor muscle electrical signals.
“It was a tough moment,” Dr. Shneider shared. “We believed this was an early warning of disease onset and that he might be at risk for severe illness.”
The neurologist offered Vierstra the same experimental treatment as his sisters to try to stave off ALS. Seizing the opportunity, Vierstra expressed, “I jumped at it.”
Every few months for the past three years, he has received spine infusions aimed at targeting and disabling the mutated gene. Though both his sisters had serious ALS complications and ultimately passed away, Vierstra believes the treatment “extended their lives.”
After a year of treatment, Vierstra found that the earlier mild abnormalities from his muscle testing had returned to normal.
Remarkably, he has yet to develop ALS and is outliving many family members. Dr. Shneider considers this research to be significant, asserting, “I think there’s real hope and a chance to make this a manageable disease, not one that is necessarily fatal.”
As Vierstra continues his work as a scientist and embraces adventures like skiing and traveling, he feels he has received a fresh chance at life. “Maybe this treatment is truly working, and I can start to think about my future in ways I couldn’t before,” he reflected.
Dr. Shneider hopes insights gained from studying familial ALS cases will eventually benefit those with non-familial forms. The ALS Center at Columbia University is also working to broaden its research beyond FUS-ALS. If anyone is interested in participating in Silence ALS, which focuses on developing personalized gene-based therapies for other rare genetic types of the disease, they can reach out via email.