An experimental gene therapy has shown promise in safely restoring hearing in individuals born with a rare type of deafness, according to recent research findings.
This study, notable for being the largest and longest trial on gene therapy for hearing loss thus far, offers compelling evidence that this method could be a viable option for those who are deaf.
“The results are truly remarkable,” said Zheng-Yi Chen, an associate scientist at Mass Eye and Ear in Boston, who was instrumental in the study, which appeared in the journal Nature. “This represents a brand-new treatment avenue for genetic hearing loss, which is quite exciting.”
The findings build on and expand the results of a smaller study from two years prior and align with outcomes from various other groups investigating similar gene therapies for different genetic forms of deafness. Notably, a treatment by Regeneron Pharmaceuticals might soon become the first gene therapy for hearing loss authorized by the FDA.
Encouraging developments in treatment
These advancements are fueling a growing interest in screening newborns for genetic types of deafness so they can receive early intervention, which tends to yield better outcomes.
“Restoring natural hearing could be a game changer in our field,” commented Dr. Lawrence Lustig, chair of Columbia University’s Department of Otolaryngology-Head & Neck Surgery.
While the specific type of deafness targeted in this study is quite rare—affecting around 50 infants annually in the U.S.—its success has prompted researchers to explore similar therapeutic approaches for other uncommon genetic hearing issues that collectively contribute to pediatric deafness.
Moreover, there are hopes that gene therapy might eventually address more prevalent types of hearing loss related to aging and high noise levels. “This is generating a lot of interest,” Lustig noted. “I know several groups planning clinical trials for these more common issues.”
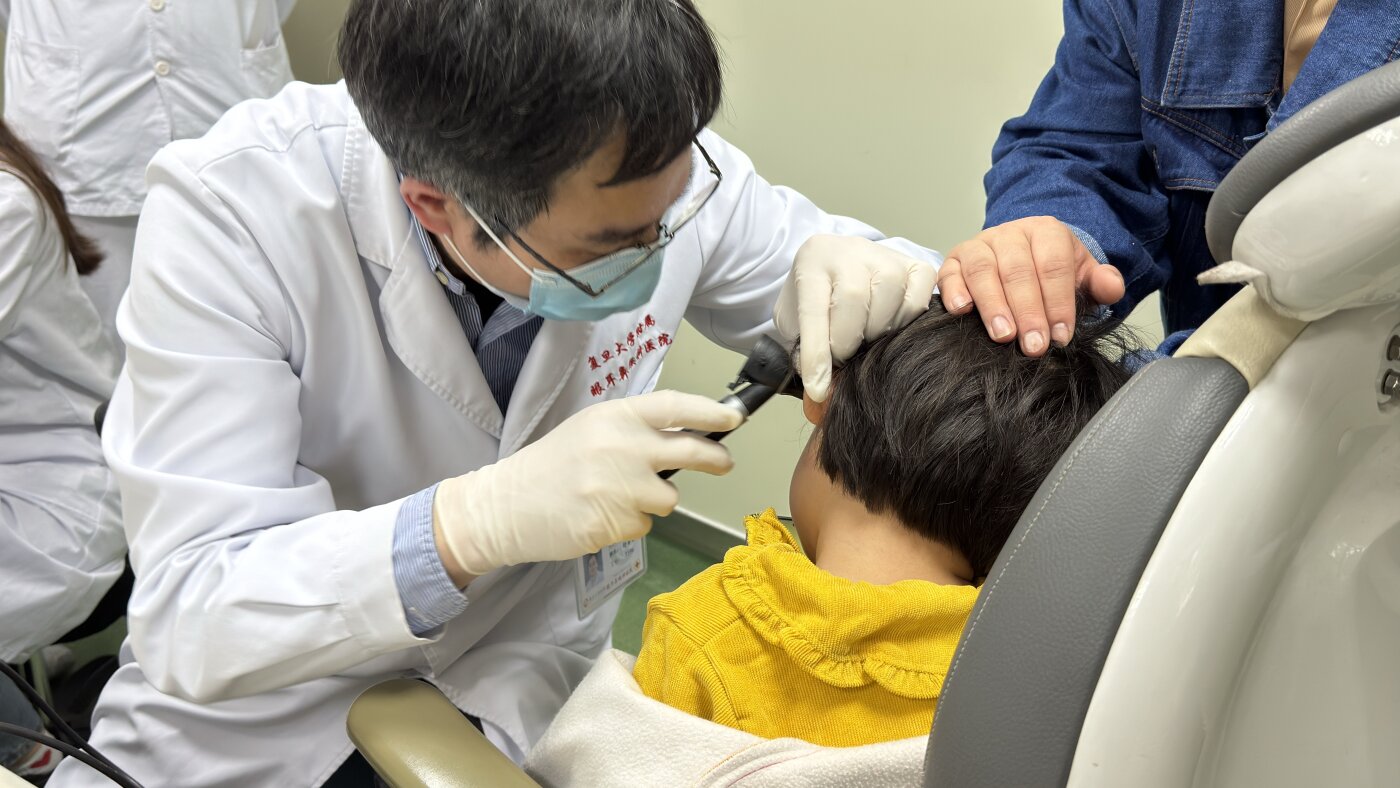
The trial at Mass Eye and Ear involved 42 individuals born with autosomal recessive deafness 9 (DFNB9), linked to mutations in the OTOF gene. This gene is crucial for the production of a protein called otoferlin, vital for hair cells in the inner ear to relay sound signals to the brain.
“Those with this mutation are born completely deaf; even loud sounds wouldn’t register,” Chen elaborated. “They wouldn’t perceive anything, even an explosion nearby.”
Delivering a healthy gene via virus
For their approach, Chen and his colleagues utilized an adenovirus to deliver a healthy version of the gene into the patients’ ears. The virus carried a modified version of the gene that allows for the production of functional otoferlin protein.
The goal was to provide the missing component in their ears so that these cells could function correctly and convert sounds into signals that the brain can interpret.
The approach appeared successful for approximately 90% of the participants, ranging from babies under a year old to adults up to 32 years in age.
According to the study, patients began to regain hearing within weeks, and their ability usually continued to improve over about six months. While the quality of hearing varied, many reached near-normal levels, and for some, benefits have persisted for over two years.
“The improvement is substantial,” Chen shared. “In some instances, patients can even hear whispers.”
Importantly, those who benefitted have started to learn how to speak, enhancing their ability to communicate with family and friends.
This research suggests that the treatment could potentially be a one-time intervention with lifetime effects, dramatically altering the lives of those affected. “I’m thrilled with these results,” Chen stated.
The therapy appears quite safe, although long-term follow-up will be necessary to ensure ongoing safety and sustained benefits.