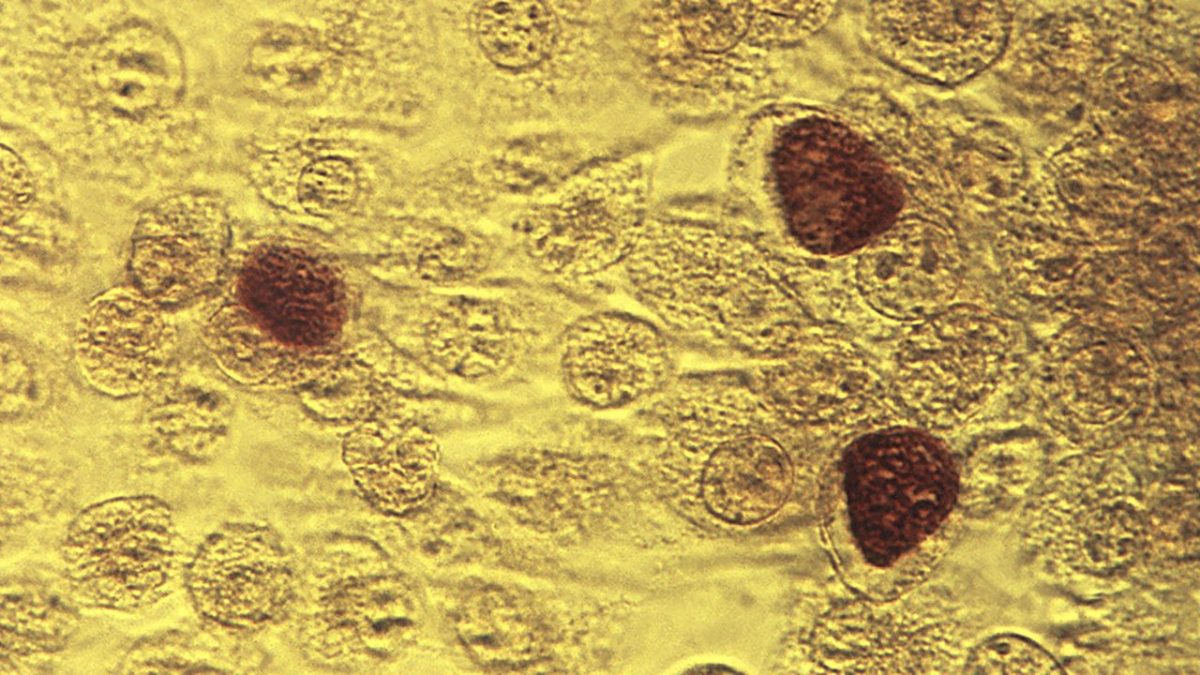
A chlamydia vaccine has shown promising results in early-stage clinical trials conducted by British and Danish researchers.
In the early stages of research, the experimental vaccine was found to be safe. The study was conducted between 2020 and 2022 in non-pregnant women and men, and none of them were infected with chlamydia. Researchers said they investigated and tested various doses of the vaccine. report It was published Thursday in The Lancet Infectious Diseases.
Participants received the vaccine or a placebo during the four-month trial period.
Researchers noted that 154 participants were screened, 65 were randomly assigned, and 60 completed the study.
Chlamydia is a common sexually transmitted disease (STD) that can cause infection between men and women. The disease can permanently damage a woman’s reproductive system, making pregnancy difficult or sometimes impossible. according to Send it to the Centers for Disease Control and Prevention (CDC).
According to the report, there is currently no vaccine available to prevent the disease, which is expected to infect more than 1.6 million people in the country by 2022. CDC.
During the trial, participants also received the vaccine through eye drops as part of an injection into the arm. Phase 2 trials will examine the shot’s effectiveness.
Questions still need to be answered in the next steps, especially since the research is in its early stages.
“Does it help prevent chlamydia infection?” said Dr. Hilary Reno, a professor of medicine at Washington University School of Medicine in St. Louis. NBC News.
“If you’re infected, that means you’re more likely to have an asymptomatic infection,” said Reno, who is also medical director of the St. Louis County Sexual Health Clinic.
“We don’t know. That will be the next stage of research,” Reno said.
Copyright 2024 Nexstar Media Inc. All rights reserved. This material may not be published, broadcast, rewritten, or redistributed.