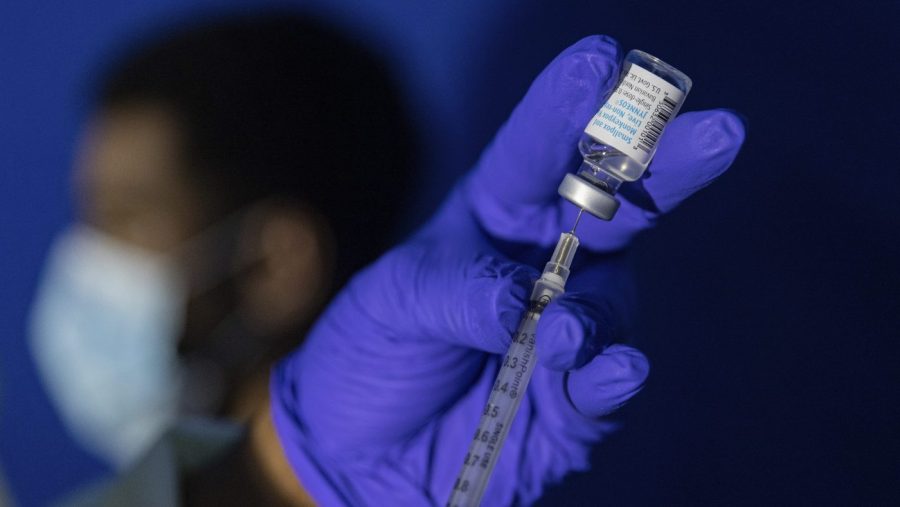
The World Health Organization announced this on Friday. It was approved The minister called the use of the MPOX vaccine an “important step” in the fight against the disease.
The move could enable national regulators to fast-track approval of the Bavarian-Nordic vaccine, making it easier to access for countries struggling to contain the outbreak.
The WHO declared MPOX a global health emergency in August following a sharp increase in cases in Central and East Africa and the rapid spread of more severe virus variants.
The Democratic Republic of the Congo is the epicenter of the MPOX epidemic in Africa, with over 5,000 cases and over 700 deaths. According to UNICEF.
“We need an urgent scale-up of procurement, donations and deployment to ensure equitable access to vaccines where they are needed most, alongside other public health measures to prevent infection, stop transmission and save lives,” WHO Director-General Tedros Adhanom Ghebreyesus said.
Bavarian Nordic The company announced that its vaccine has been approved for immunization against smallpox, measles, and related orthopoxviruses in adults 18 years of age and older.
But in a statement, WHO added that the vaccine may be given “off-label” to infants, children, adolescents, pregnant women and immunocompromised people if the benefits of the vaccine outweigh the risks.
Children are particularly vulnerable to MPOX because their immune systems are still developing and they are at higher risk of developing severe MPOX. According to the WHO.
Symptoms of this disease in children can be similar to many other common childhood illnesses, such as chickenpox and other viral infections.
The vaccine is typically administered to adults in two doses, four weeks apart, but the WHO recommends a single dose in outbreak situations where vaccine supplies are limited.
Bayer Nordic's MPOX vaccine has been in use since 2022, when the U.S. Food and Drug Administration and the European Medicines Agency issued emergency use authorization for the drug to combat a different MPOX strain.
According to the WHO, data shows that one dose of the MVA-BN vaccine administered before infection is approximately 76% effective in preventing MPOX infection, and two doses are 82% effective.