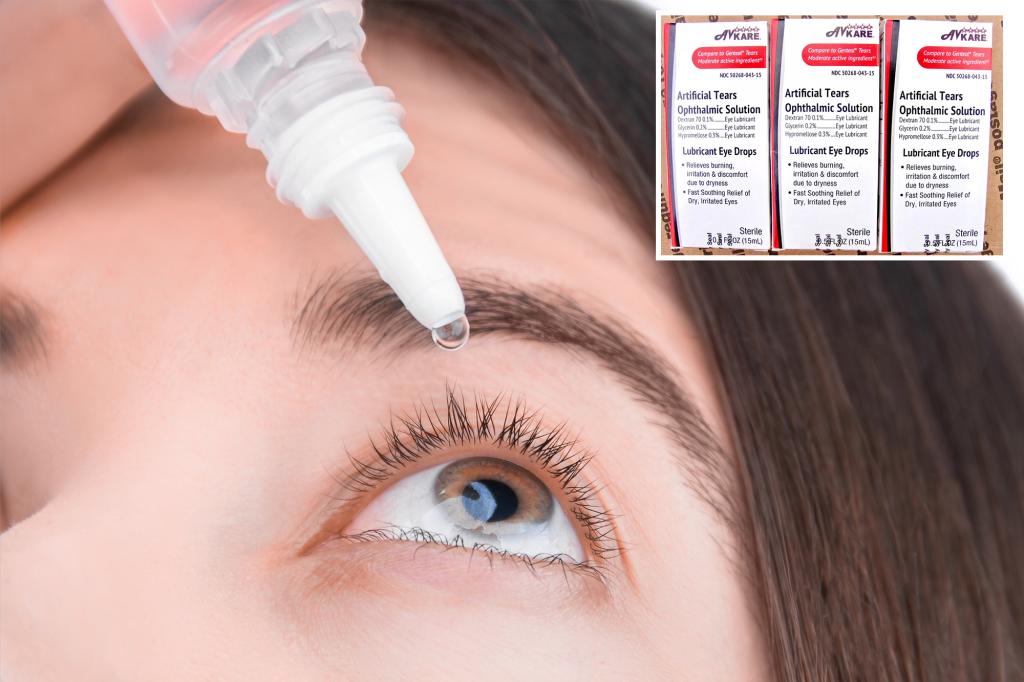
In a recent report from the FDA, it was revealed that 76,000 eye drop units are under recall, initiated by the drug consulting firm BRS Analytics Services.
This emergency measure came about after the FDA’s audit uncovered several compliance deviations at the original manufacturer, Avkare. You can find more details about the recall through their official site.
The affected eye drops, intended for alleviating itchy, dry eyes, were flagged due to concerns over what the FDA referred to as a “lack of infertility guarantees.”
The potential health risks tied to these eye drops currently remain unclear, but the company, Avkare, has advised customers to stop using the impacted products immediately.
Recalled Eye Products:
- NDC#50268-043-15 Artificial Tear Solution
- NDC #50268-066-15 Carboxymethylcellulose Sodium Ophthalmic Gel 1%
- NDC#50268-068-15 Carboxymethylcellulose Sodium Eye Solution
- NDC#50268-126-15 Lubricant Ophthalmic Solution
- NDC#50268-678-15 Polyvinyl Alcohol Ophthalmic Solution
The recalled items were shipped between May 2023 and April 2025, with expiration dates stretching from April 2025 to March 2027.
Consumers looking to return the recalled products can do so by sending them back to Avkare for a full refund, including shipping costs. To proceed, you’ll need to fill out a specific form found on Avkare’s website and send it via fax or email to obtain your return slip.