Blood Tests for Alzheimer’s Diagnosis Approved
In a significant move, U.S. health officials approved the first blood test aimed at diagnosing Alzheimer’s disease, with the hope of identifying potential patients. This test can assist doctors in differentiating whether a patient’s memory issues stem from Alzheimer’s or other medical conditions that affect cognitive function. The FDA has authorized its use for individuals over 55 who exhibit early signs of the disease.
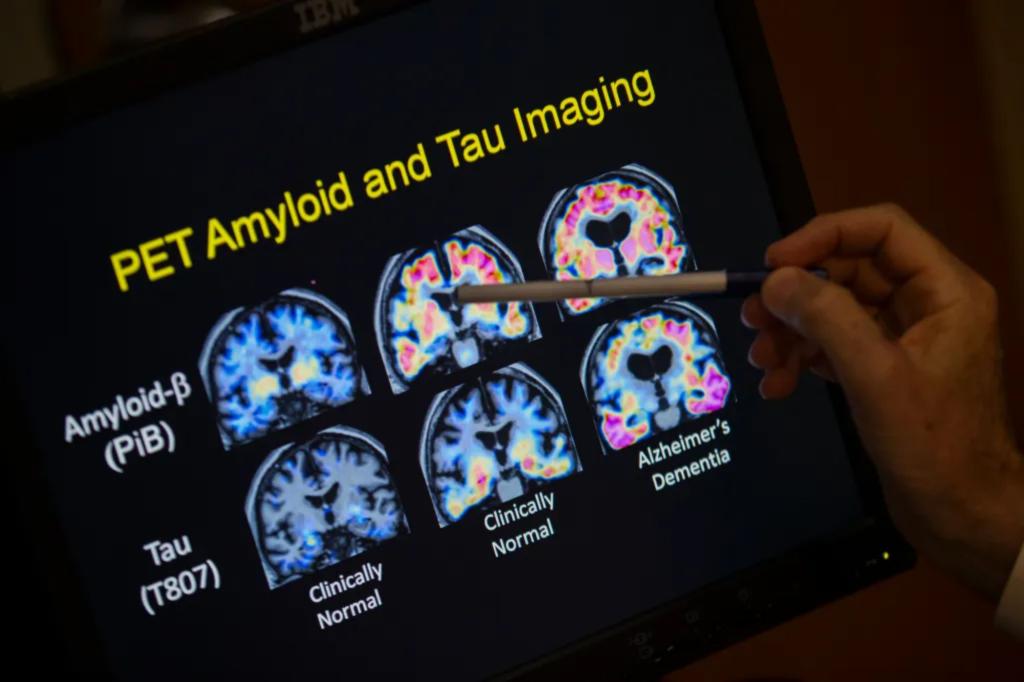
Alzheimer’s disease is a widespread issue, affecting over 6 million people in the U.S. and millions more globally. The new test, developed by Fujirebio Diagnostics, Inc., detects beta amyloid plaques in the brain, which are key indicators of Alzheimer’s. Previously, the only available FDA-approved methods for identifying amyloid involved invasive spinal fluid testing or costly PET scans.
The convenience and lower cost of these blood tests could significantly broaden the reach of two new medications, Leqembi and Kisunla, which have demonstrated the ability to modestly slow Alzheimer’s progression by eliminating amyloid in the brain. Doctors will need to confirm the presence of plaques before prescribing these medications that require regular IV infusions.
Dr. Michelle Tarber, PhD, from the FDA’s Device Center, remarked, “Today’s clearance is an important step in diagnosing Alzheimer’s disease, making it potentially easier and more accessible for U.S. patients in the early stages.” However, despite advancements, many specialized hospitals and research institutions have been developing their own amyloid tests without FDA review, often leading to inconsistencies in reliability and coverage by insurance.
Several major companies in diagnostics and pharmaceuticals, like Roche and Eli Lilly, are also working on tests that could receive FDA approval in the future. It’s important to note that these blood tests can only be ordered by a physician and are not intended for asymptomatic individuals.