Back in 1958, a theory about vitamin B1—or thiamine—was largely disregarded by scientists. The hypothesis suggested that this essential vitamin, crucial for metabolic functions, could form a very reactive intermediate during specific biochemical reactions. Surprisingly, recent studies indicate that this notion might actually be correct.
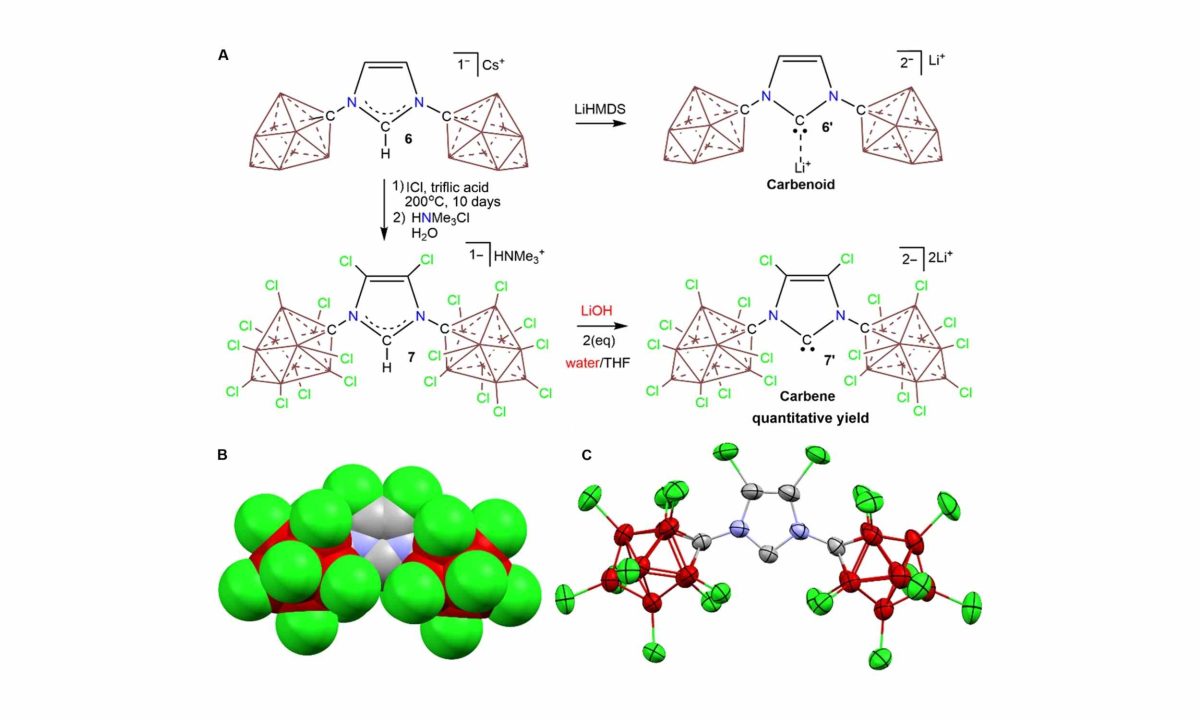
Led by Prof. Vincent Lavallo from the University of California, Riverside (UCR), researchers have demonstrated that vitamin B1 can indeed generate a reactive species known as a carbene, even in aqueous environments.
Understanding vitamin B1 and carbenes
Carbenes are characterized by having only six valence electrons on their carbon atom. This makes them highly reactive, often disappearing almost immediately after formation. Traditionally, it was believed that water would destabilize these molecules before any thorough examination could occur.
“For the first time, we’ve observed a stable carbene in water. Many thought this was absurd, but it seems that Breslow was onto something,” remarked Lavallo.
Thiamine plays an integral role in fundamental bodily functions, particularly the breakdown of sugars for energy.
Why so much doubt for so long?
Historically, carbenes were deemed too unstable to exist in water, as they react so quickly. Even tiny amounts of moisture in the air could lead to their destruction before scientists could observe them.
Most early efforts to study carbenes in water resulted in failure; they either vanished or decomposed into other molecules, which solidified the belief that they couldn’t survive in aqueous environments. This skepticism was so strong that researchers chose dry, oxygen-free conditions for their experiments, making the idea of them existing in water seem far-fetched.
A fresh approach to working in water
Thinking about a carbene forming in water seemed impossible because water typically reacts with such electron-deficient carbon structures right away. Dr. Varun Raviprolu, a postdoctoral researcher at UCLA, explained, “We were actually focused on exploring their chemistry, not trying to validate a historical theory, yet our work ended up exactly confirming what Breslow proposed years ago.”
To achieve this, Raviprolu and his team developed a special molecular shield to protect the carbene from interacting with water, effectively preventing immediate breakdown.
1958 theory turned out to be true
Chemist Ronald Breslow theorized long ago that thiamine might convert to a carbene during certain reactions in the body, but no one could provide conclusive evidence. The new findings clearly show that, under specific conditions, vitamin B1 can stabilize as a carbene form, making it observable using advanced techniques like nuclear magnetic resonance.
This newfound stability helps clarify how vital transformations happen within living cells. Researchers suggest that the body might be forming brief carbene intermediates during ordinary metabolic processes.
Eco-friendly B1 manufacturing
Carbenes can function as ligands in metal-based catalysts, extensively used for synthesizing various substances. Their presence in water could lead to more environmentally friendly methods since there’s less reliance on hazardous solvents.
“Water is an ideal solvent—it’s plentiful, non-toxic, and eco-friendly. If we get these powerful catalysts to function in water, it’s a significant advancement toward greener chemistry,” noted Raviprolu.
Such catalysts have the potential to revolutionize how pharmaceuticals and common materials are produced, especially since many industrial reactions are notorious for creating chemical waste.
By leveraging water-based techniques, there’s a hope to lower costs and minimize safety risks associated with flammable liquids.
Mimicking life’s chemistry in the lab
Living cells depend on intricate, aqueous reactions to function smoothly. Many of these processes involve rapid, reactive intermediates that are challenging to detect or replicate in laboratory settings.
The successful stabilization of a molecule that reflects this behavior could allow researchers to more accurately recreate cellular chemistry, enhancing their understanding of enzyme function. It might even introduce new paths for repairing or reprogramming these reactions in medicine.
Carbenes are frequently examined for their capacity to imitate components of enzyme action. Now that scientists can fabricate them in water, they may discover methods to control them for specific biological tasks.
This could eventually shift how enzyme-inspired drugs are designed or materials that react solely in conditions akin to those within human cells.
Next steps with vitamin B1
Establishing that carbene intermediates can endure in water unravels a mystery surrounding vitamin B1’s role. More so, it sparks questions about other ephemeral molecules that might exist in living organisms.
Lavallo and his team are eager to explore whether similar techniques could unveil additional hidden agents involved in significant biological reactions.
“What seems implausible today could become possible tomorrow with ongoing scientific investment,” indicated Raviprolu.
This discovery serves as a reminder that no theory should be dismissed as implausible without comprehensive investigation.
The details are published in Science Advances.