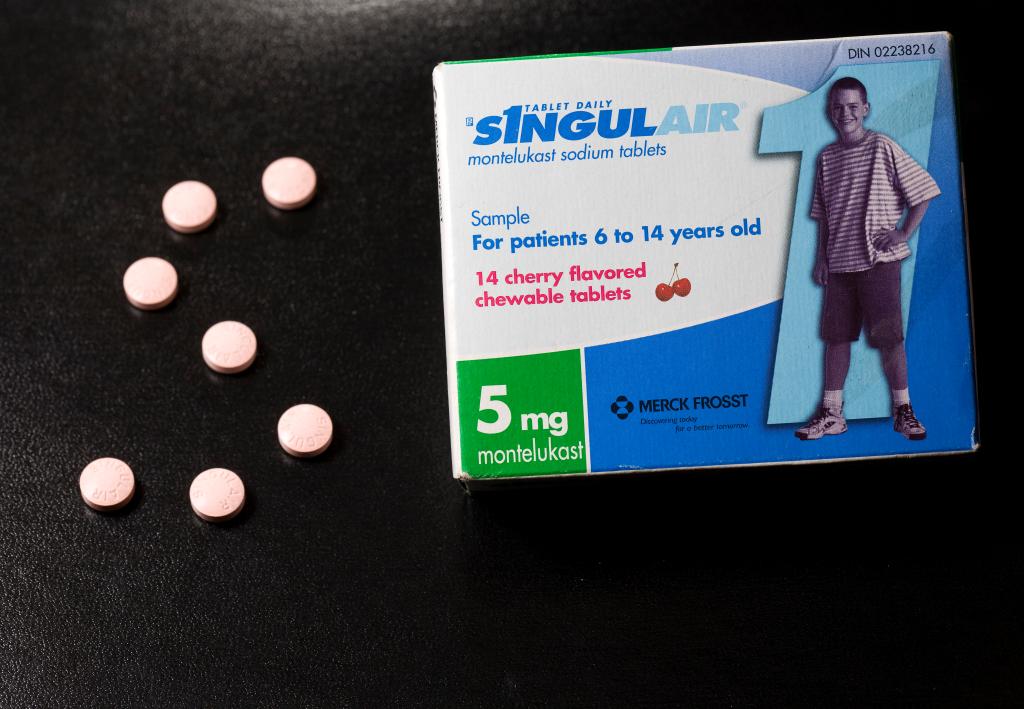
The Food and Drug Administration (FDA) issued a warning against commonly prescribed asthma medications in 2020, but millions of people are still taking them.
A black box warning (also known as a box warning) Published by FDA in March 2020 Regarding the mental health side effects of Singulair, also known by its generic name, montelukast — Asthma and allergy medication 25 years ago.
According to , a boxed warning is the highest safety warning that the FDA can assign to a drug. National Institutes of Health (NIH (National Institutes of Health).
The warning was in response to decades of escalating concerns about aggression, agitation and even suicidal thoughts. Although the FDA warned medical groups, it did not require physicians to be educated about the drug's side effects.
This warning was issued mainly by doctors prescribing the drug to convey information to patients, but it appears that very few doctors were actually informed.
Despite the stark warnings, 12 million people in the U.S. will still be prescribed the drug by 2022, including 1.6 million children. The New York Times reported.
Experts stressed that children are particularly susceptible to side effects.
The outlet obtained the data from healthcare analytics firm Komodo Health, based on claims submitted to Medicaid and Medicare, as well as claims submitted to private insurance companies.
singlair manufacturerMerck & Co. sells some products organonJune 2021, including the singular (monelukast sodium).
“Nothing is more important to Organon than the safety of our medicines and the people who use them. We take reports of adverse events seriously and, as with all of our medicines, we continue to ensure the safety of Singulair. Organon said in a statement to the Post.
“We remain confident in the efficacy and safety profile of SINGULAIR when used in accordance with the FDA-approved prescribing information. SINGULAIR's product label contains information on SINGULAIR's benefits, risks, and reported side effects. Contains appropriate information.”
“Organon is confident that we have communicated complete and appropriate information about Singulair to healthcare providers and patients to ensure the safe and effective use of the drug in accordance with the prescribing information,” the representative added, noting that patients should be aware of any concerns. Providers who have indicated that you should discuss the matter with your healthcare provider.
The fact that the drug is still being prescribed in high concentrations, despite increasing reports from affected patients, is a warning to doctors and patients who often don't read the fine print on drug labels. This is raising concerns about whether enough has been received.
One woman, Nicole Sims, told the Times that when her 6-year-old son started having nightmares and hallucinations of the woman in the window and told him he wanted to die, she turned to the Internet for answers. reported. She claimed she first heard of the FDA warning through an internet search.
Sims also came across a Facebook support group. Montelukast (Singulair) Side Effects Support and Discussion Group — More than 20,000 members share their experiences with drug side effects.
The members discovered the link between the side effects and the drug with the help of their peers rather than doctors.
“This is a mental health crisis that no one is aware of,” Anna Maria Rosenberg, the group's administrator, told the Times.
Dr. Reshma Ramachandran, an assistant professor at Yale University and a family physician, told the Journal that she's concerned that few doctors know about the warning, and that it's not getting through the channels that doctors are aware of.
Ramachandran added that the FDA “needs to do more through direct communication with clinicians and more proactive channels.”