Antibiotics really transformed the landscape of medicine. They made once-fatal infections manageable and have been integral to modern surgeries, cancer treatments, and the survival of premature infants.
However, we seem to be hitting a wall. Around the globe, bacteria and fungi are evolving more quickly than our treatments can keep up with. Infections persist, and drugs that were once considered the last line of defense are failing.
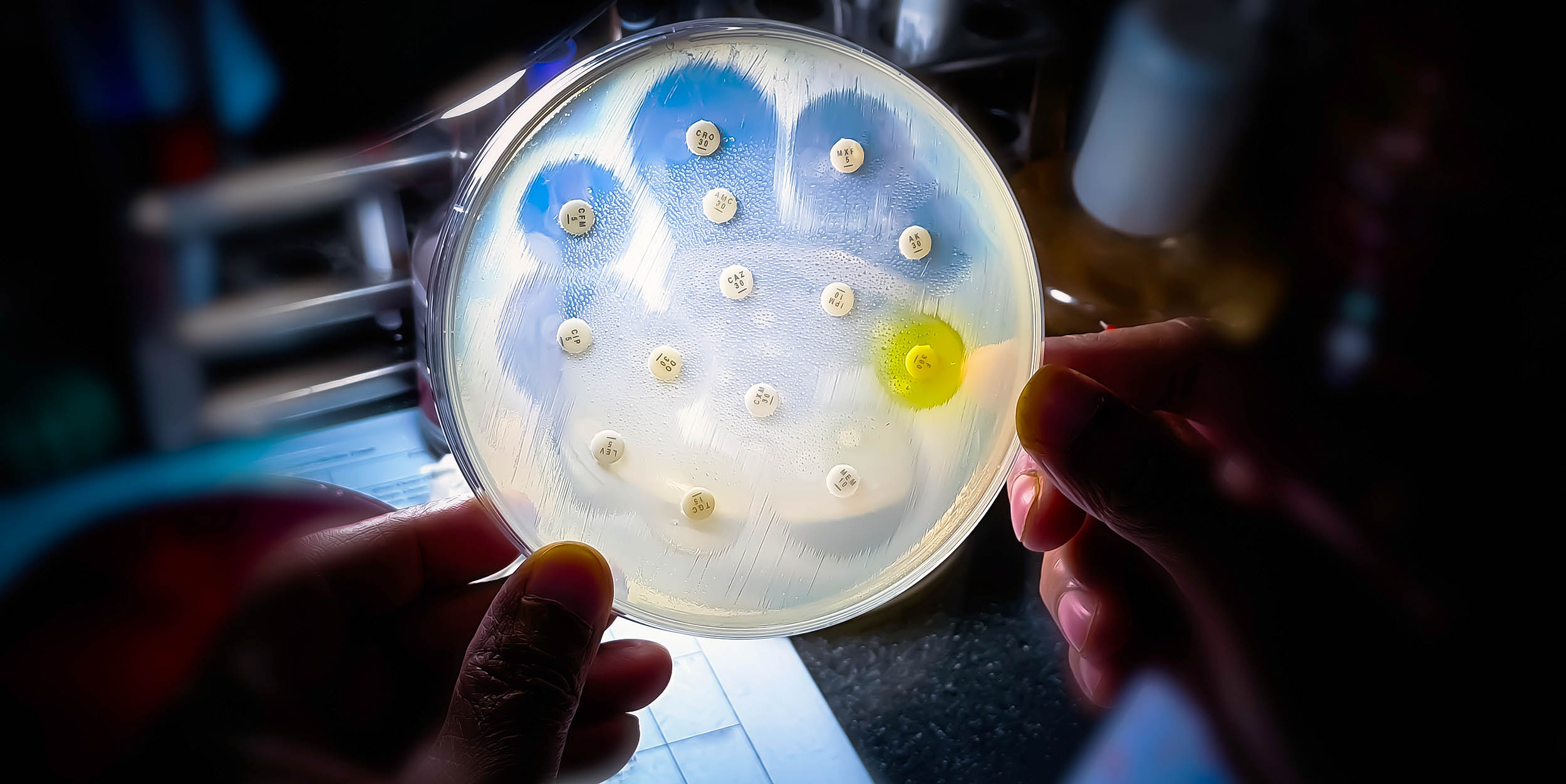
A recent comprehensive review led by Jilin University and Peking Union Medical College Hospital has gathered crucial data from hospital labs and national surveillance efforts, showing that antimicrobial resistance (AMR) is not a future concern but a current crisis. It varies by region and is influenced by the workings of local health systems. On the bright side, we have a good understanding of how to slow down this resistance, and we can act on that knowledge now.
A global problem with a local accent
The rise of resistance isn’t uniform everywhere. It reflects local policies, practices, and everyday habits.
In parts of Asia, for instance, strains of E. coli and Klebsiella that produce β-lactamases—enzymes that break down key antibiotics—are prevalent. Meanwhile, in Europe and the Americas, resistance to carbapenems, which are supposed to be our safety net, is steadily increasing.
Some pathogens found in intensive care units, like Acinetobacter baumannii and Pseudomonas aeruginosa, exhibit over 70% resistance in specific areas. Conversely, several Northern European countries maintain low resistance rates, primarily due to stringent prescribing practices, effective infection control, and proactive monitoring. Same germs, vastly different outcomes. It really highlights the importance of systems.
It’s not just bacteria
Fungi aren’t sitting this fight out either. Candida auris, a resilient yeast found in hospitals, has gained resistance to several antifungal medications and can spread rapidly if not properly managed.
In the environment, Aspergillus fumigatus is also developing resistance to azole treatments, partly due to the widespread use of the same chemical class in agriculture. This connection between farms and medical facilities really exemplifies why AMR is a One Health issue: human health, animal health, and the environment are interconnected.
Outsmarting antibiotic resistance
The review outlines the various strategies microbes employ. β-lactamases and carbapenemases disable antibiotics, efflux pumps eject them from cells, and minor genetic mutations alter drug targets so that our medications can’t attach properly.
Resistance genes can jump between bacteria via mobile DNA, spreading through wastewater, animal hosts, and even travelers. But none of this is set in stone. It means that relying on broad-spectrum drugs is not a viable long-term strategy. Finding the right medication, dosage, and treatment length tailored to the specific infection is key.
Precision beats panic
Broad-spectrum treatment based on a “better safe than sorry” mentality contributed to this problem. But precision medicine offers a solution. This involves using rapid diagnostics and, when feasible, genetic testing to pinpoint pathogens and their resistance markers.
Dosing should be based on how the drug is metabolized and its effects on the bacteria, rather than adopting a one-size-fits-all approach. It might also include combining β-lactams with specific β-lactamase inhibitors or strategic regimens that allow us to revive older medications like polymyxins when other options are exhausted. Swift action is essential when lab results come in.
Monitoring antibiotic resistance
If you’re curious about why some nations have low resistance rates while others face alarming levels, look at their policies.
Effective stewardship programs that necessitate cultures prior to prescribing carbapenems, regulations to restrict antibiotic availability without prescriptions, and investments in hygiene and isolation protocols lead to better resistance outcomes over time. The review advocates for similar foundational efforts: national surveillance that communicates with hospitals and farms, stringent prescription regulations, and clear incentives for using fewer, more targeted drugs when appropriate.
Agriculture and the environment
Antibiotics and antifungals aren’t confined to clinics. The preventive use in livestock and routine fungicides can select for resistant organisms that return to humans.
Improving livestock management, developing vaccines for animals, employing targeted treatments instead of blanket applications, and reducing agricultural substances that contribute to medical cross-resistance are practical measures to safeguard both food production systems and healthcare. Additionally, monitoring wastewater and enhancing sanitation can intercept additional pathways for resistant gene transmission.
Drugs and better tests are needed
We can’t just focus on preservation without innovation. New antibiotics and antifungals are essential, especially against stubborn Gram-negative bacteria, along with next-generation β-lactamase inhibitors and agents that disrupt pumps or biofilms.
Equally pressing is the need for improved, quicker diagnostics. Every day we can shorten empirical treatments reduces harm to patients’ microbiomes and minimizes the risk of prompting resistance. AI-driven decision tools are beginning to assist doctors in making better, faster choices.
The human part of the puzzle
However, none of this is effective without human involvement. Clinicians require adequate time and tools to determine the appropriate tests and modify treatments. Pharmacists and stewardship teams must have the authority to intervene when necessary.
Patients also need clear communication: antibiotics won’t treat viral infections; leftover medications shouldn’t be kept as mementos; and stopping treatment early is dangerous. Public messaging should be clear and easy to understand, ensuring everyone can adhere without needing a medical background.
A slow-moving pandemic
AMR isn’t a sudden event; it’s a gradual rise. While that sounds negative if we remain inactive, it offers hope if we take measures—after all, tides can be anticipated and mitigated.
“AMR represents a slow-moving pandemic,” explained Xuesong Xu, the study’s lead author. “Our review indicates that resistance patterns are ever-changing and influenced by human actions, healthcare systems, and environmental issues.”
“No nation can tackle this crisis alone. A unified One Health strategy is crucial, linking human health, agriculture, and ecology. Strengthening surveillance, optimizing antibiotic use, and incentivizing innovation are vital to prevent a future where routine infections become life-threatening again.”
The roadmap is straightforward: enhance surveillance, prescribe with care, invest in diagnostics and new medications, reform agricultural practices, and educate the public to view antibiotics as a precious, limited resource.
By consistently implementing these strategies, we don’t just buy time; we rebuild the essential safety net that modern healthcare relies upon.
The findings are published in the Medical Journal of Peking Union Medical College Hospital.