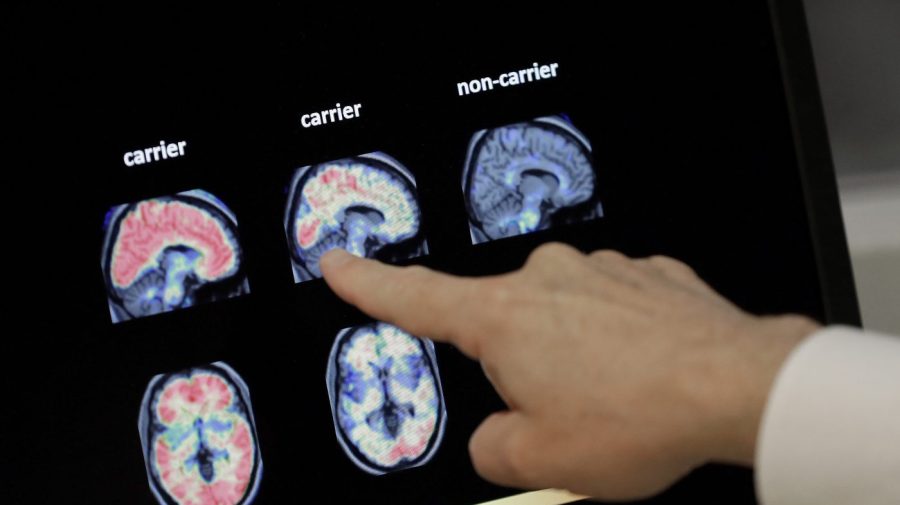
On Friday, the Food and Drug Administration (FDA) revealed a new test designed to diagnose Alzheimer’s disease in adults. This is the first of its kind that uses blood samples to determine the presence of the illness.
The test is known as the “Lumipulse g ptau217/ß-amyloid 1-42 plasma ratio” and was developed by Fujirebio Diagnostics, Inc., based in Japan. The accumulation of amyloid plaques in the brain is a defining characteristic of Alzheimer’s disease.
Fujirebio submitted the necessary regulatory applications to the FDA for evaluation back in September. This marks the first blood-based diagnostic test for Alzheimer’s available in the U.S.
“Alzheimer’s disease impacts more individuals than breast and prostate cancer combined,” said FDA Commissioner Martin McCurry in a statement. “Currently, around 10% of those over 65 have Alzheimer’s, and projections suggest that by 2050, that number could double. We hope that innovative medical products like this will benefit our patients.”
The FDA also highlighted that blood draws for this test are less invasive compared to other existing options. They noted that about 92% of participants who tested positive for Alzheimer’s were confirmed to have amyloid plaques through either PET scans or cerebrospinal fluid examinations. Fewer than 20% of participants received ambiguous results.
The Alzheimer’s Disease Association reports that over 7 million Americans are affected by this condition, with projections estimating that figure will rise to nearly 13 million by 2050. Currently, one in ten Americans aged 65 and older has Alzheimer’s disease.
The FDA stated, “These findings indicate that new blood tests can accurately predict whether amyloid pathology associated with Alzheimer’s is present or absent in patients with cognitive impairment. This test is meant for patients showing signs of cognitive decline and should be interpreted alongside clinical information from other sources.”
Howard Fillit, co-founder and chief science officer of the Alzheimer’s Drug Discovery Foundation, referred to the test as “a significant milestone for both patients and clinicians.”
“The ability to diagnose Alzheimer’s disease through a simple blood test, similar to cholesterol testing, could be transformative, enabling more patients to access treatments that may slow down or even prevent the disease,” Fillit remarked.
“This represents a new era in Alzheimer’s research, where advancements in science and technology are converging to create more accessible and affordable diagnostic tools, setting the stage for further regulatory approvals,” he added.