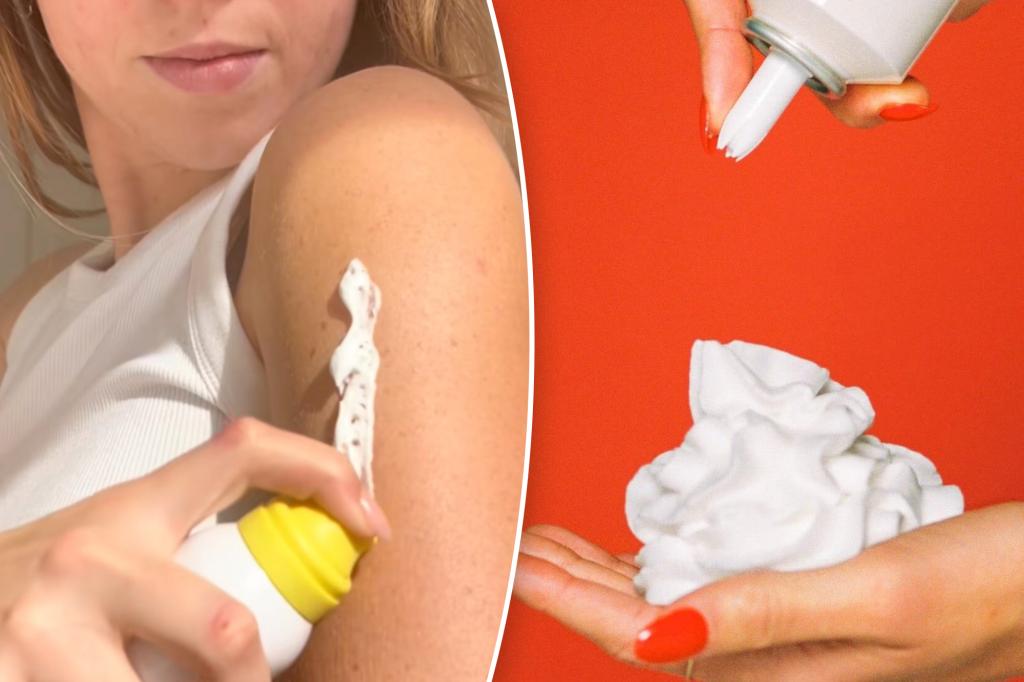
This summer, some of the most popular beauty products have raised concerns.
The Food and Drug Administration (FDA) issued a warning about mousse-style sunscreens, suggesting they might not be effective. I think they posted about it on X recently.
The alert follows a series of warning letters sent to brands like Super Goop!, Vacation Inc., and others for selling whipped, mousse, and foam sunscreens that don’t comply with federal regulations.
The FDA’s warning highlights that these products might not meet the criteria for over-the-counter drugs in the U.S., which includes strict rules about formulations.
Generally, the FDA states that sunscreens should only be sold as oils, lotions, creams, gels, butters, pastes, ointments, sticks, sprays, or powders. Companies need to submit new drug applications with adequate evidence to support their safety and effectiveness to sell products legally.
The agency pointed out that there isn’t any valid FDA-approved application for these mousse formulations.
The warning also targeted Vacation Inc.’s “Classic Whip Sunscreen,” packaged to resemble a whipped cream can and marketed as a “skin dessert.” It raises the question of whether such packaging might confuse consumers into thinking it’s food.
That said, just because the FDA raised concerns doesn’t necessarily mean these fluffy sunscreens are ineffective. A spokesperson for Kalani Sunwear commented that the warning didn’t imply that these products lack burn-blocking abilities or are unsafe. They noted that their Sun Mousse SPF 50 is produced to high standards in Sweden.
A representative for Super Goop! emphasized that the FDA’s focus was on labeling rather than the product’s safety or effectiveness, and they’re actively collaborating with the FDA to address the issue.
Despite the warnings, some experts seem skeptical of the mousse format. For example, Ava Perkins, a cosmetic chemist, explained that SPF is determined by applying a specific amount of the product, and the incorporation of air in these mousse sunscreens could complicate measurements.
The FDA’s letter, dated August 6th, gives the companies 15 business days to either resolve the issues or demonstrate compliance with the law. Kalani Sunwear has started taking action by temporarily removing their mousse-style sunscreen from their U.S. website to ensure they meet regulations.
This post is looking for responses from Vacation, Kalani Sunwear, K&Care Organics, and Fallien Cosmeceuticals.