Cells
Peripheral blood mononuclear cells (PBMCs) were extracted from leukocyte cones sourced from healthy donors via density centrifugation, utilizing Lympho 24+ Spin medium and pluriMate II tubes. These PBMCs were then cryopreserved in a solution containing 10% DMSO in FCS. CD4+ T cells were isolated through a negative selection process that used a specific isolation kit, before being cultured in a medium enriched with RPMI1460, FCS, penicillin-streptomycin, GlutaMax, and IL-2. A cocktail of biotin antibodies was employed for CD25/CD69 depletion, ensuring that the expressing cells remained in the negative fraction. Their purity was then confirmed through flow cytometry. The isolated CD4 T cells underwent activation in Nunclon Delta flasks with a plate-bound anti-CD3 antibody and soluble anti-CD28 antibody. After three days of activation, they were rested in fresh medium for two additional days prior to infection or co-culture. Meanwhile, Jurkat T cell lines were maintained in complete RPMI medium. Other cell lines, such as HEK293T/17 and HeLa-TZMbl, were cultured using DMEM GlutaMax with FCS and penicillin-streptomycin.
Plasmids
The pNL4.3 plasmid was a contribution from M. Martin. Variants of this plasmid were provided by others. For instance, pNL4.3 ΔEnv and pNL4.3 ΔVpr came from R. Sloan, while additional plasmids were shared by G. Towers and others from NIH sources. Certain specialized plasmids were also derived from various contributors, with coding for specific proteins noted. The modifications to plasmids, including mutagenesis and fusion constructs, were made using standard techniques, such as the QuickChange kit and selected primers.
Viruses
Viruses were generated via transfection of 293T cells with HIV-1 plasmids using Fugene6. Specific viral traits, such as pseudotyping, were achieved through co-transfection with variants of the HIV envelope. Efforts were made to quantify virus production, and different methods, including RT activity measurement or infectivity assays, were employed for this purpose.
HIV-1 infections, VS priming and bead priming
Donor cell infection
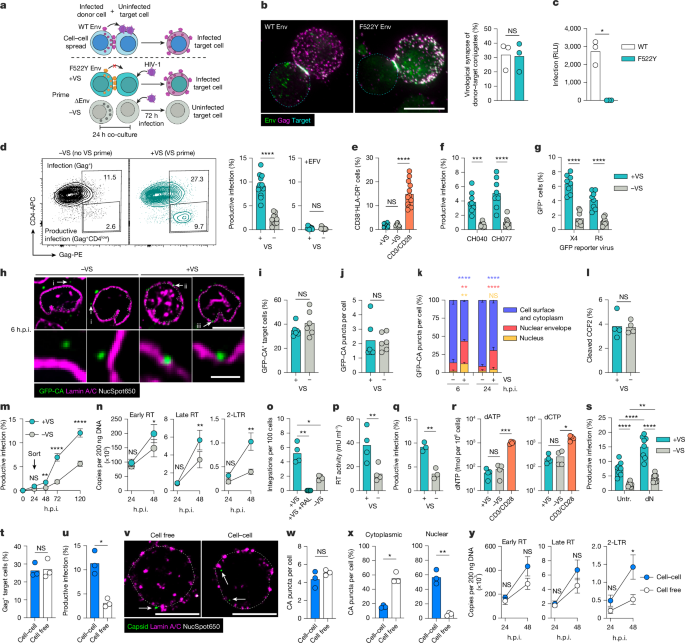
Activated CD4 T cells were infected on day five post-activation, using spinoculation with Env-pseudotyped viruses. Jurkat T cells were similarly infected but via gravity methods. Confirmation of infection was conducted using flow cytometry.
VS priming
Target T cells were pre-labeled and then combined with donor T cells, challenged with various HIV-1 strains. Subsequent infection measures were recorded through flow cytometry.
Bead priming
Beads coated with specific antibodies or Env were prepared for priming target T cells, followed by a standard infection procedure.
CCS assay
Infection levels were assessed using both donor and target cells infused with viral content, monitored for specific cellular responses. These were complemented with quantitative PCR analysis for thorough data examination.
Jurkat 1G5 CCS assay
For further study, Jurkat T cells were co-cultured to evaluate luciferase expression following HIV-1 infection.
Blam-Vpr assay
Utilization of the NL4.3 Blam-Vpr virus allowed for measuring β-lactamase activity in infected cells.
Inhibitors
Specific inhibitors were applied to cells pre-treatment and during viral exposure to assess their effect on infection and signaling pathways.
T cell signalling
For signaling measurements, T cells were stained and analyzed under specific conditions to capture response dynamics post-activation and in the presence of beads.
Flow cytometry
Cell samples were processed to evaluate surface and intracellular marker expressions. Staining techniques were employed to ensure detailed profiling of T cell responses and dynamics.
Live-cell sorting
Following co-culture, live sorting was executed to isolate T cell populations based on specific labels, with high purity confirmed through subsequent flow cytometry analysis.
CDK1 mRNA electroporation
Specific CDK1 sequences were prepared, followed by electroporation into T cells to analyze expression levels through various assays.
CDK1 CRISPR–Cas9 knockout
The knockout methodology allowed for targeting CDK1 genes, monitored post-electroporation via flow cytometry.
RT–qPCR analysis
The quantification of viral products from cell DNA was performed utilizing established techniques, with a focus on detecting early and late RT products.
Quantification of cellular dNTPs
The levels of dNTPs were measured with qPCR to assess their implications on T cells during HIV-1 infection.
Immunoblotting
Sample processing for immunoblotting involved lysing cells in specialized buffers and utilizing antibodies for specific proteins to assess their expressions.
dSTORM imaging
This advanced imaging technique was employed to analyze the interactions at a cellular level, focusing on labeling and capturing dynamic processes.
dSTORM analysis
Nucleoporin interactions with HIV-1 were precisely quantified, utilizing fitting calculations for detailed assessments.
MS analysis
Sample preparation
Cells underwent specific treatments before being processed for proteomics analysis, including steps for digestion and purification.
Phosphopeptide enrichment
This protocol allowed for the selective enrichment of phosphopeptides for downstream applications.
LC–MS
Various parameters were optimized for liquid chromatography coupled with mass spectrometry to analyze peptide samples effectively.
Data analysis
Comprehensive analysis of the proteomics data utilized R for statistical processing and relevant corrections.
Statistical analysis
A variety of statistical evaluations were employed to draw conclusions on experimental data, with details provided on methodologies used.
Reporting summary
Additional research design information can be accessed via the linked reporting summary.