the patient Hard-headed person syndrome We are one step closer to accessing new treatments.
Kiberna Therapeutics announced on Monday that its new drug, KYV-101, has been granted Regenerative Medicine Advanced Therapy (RMAT) designation by the U.S. Food and Drug Administration (FDA).
Drugs eligible for RMAT designation are those “intended to treat, ameliorate, ameliorate, or cure a serious or critical disease.” Life-threatening illness According to the FDA website, a drug is effective for a disease or condition “or condition” if “preliminary clinical evidence indicates that the drug may address an unmet medical need for such disease or condition.”
Stiff-Person Syndrome Patient Explains What It’s Like Living with a Rare Disease
One benefit of RMAT, according to the company, is that it allows Kyverna to work closely with the FDA to help expedite development, review and approval.
According to a company press release, the FDA’s decision is based on positive results from clinical trials in patients.
Carrie Robinette, left, was diagnosed with stiff-person syndrome in 2023. The FDA granted KYV-101 regenerative medicine advanced therapy (RMAT) designation on Monday. (Carrie Robinette, iStock)
Stiff-person syndrome, a rare neurological disorder, affects only one or two in every million people. Singer Celine Dion.
The disease can have devastating effects, causing muscle stiffness, pain, spasms and loss of movement.
Like Celine Dion, Pennsylvania man is battling stubborn syndrome ‘with all his might’
Kiberna Therapeutics is Emeryville, CaliforniaAccording to the company, it has developed a novel CAR-T cell therapy, KYV-101, with the aim of “resetting” the immune systems of patients with autoimmune diseases.
“As a physician dedicated to optimizing the diagnosis and treatment of patients with autoimmune diseases, Neurological disorders“Through our collaborations with leading researchers in the field and FDA oversight support, we are pleased to witness and contribute to advances in treatments and patient outcomes for stiff-person syndrome,” Amanda Pique, MD, director of the Autoimmune Neurology Program at CU Anschutz Medical Campus in Aurora, Colorado, told Fox News Digital in an email.

Celine Dion announced to the world in December 2022 her diagnosis with Stiff Person Syndrome. (Edward Barcello/GC Images/Getty Images)
Piqué was not involved in the development of KYV-101 but served on one of the company’s medical advisory committees.
Kyverna CEO, Dr. Peter Maag, also commented on the announcement:
“This RMAT designation means that the science agrees that expediting this treatment has the potential to save lives.”
“Kiverna is committed to supporting patients and their clinical unmet needs, and is building patient-centric clinical trials to provide access to the latest medical advances in the field of CAR-T for autoimmune diseases,” Maag said in a written statement to Fox News Digital.
“We are proud to investigate the potential for safe and effective Long-term treatment This could potentially lead to meaningful and durable remission for patients suffering from autoimmune diseases like SPS.”
Click here to sign up for our Health Newsletter
Patients, too, reacted with excitement to the news.
“From the moment I saw how well Kaiverna’s treatment helped SPS patients in Germany, it’s been my dream that FDA approval would come soon so we can all benefit,” Carrie Robinette, 45, of San Diego, California, told Fox News Digital.

According to the FDA’s website, a drug is eligible for RMAT designation if it is “intended to treat, ameliorate, ameliorate, or cure a serious or life-threatening disease or condition” and if “preliminary clinical evidence indicates that the drug has the potential to address an unmet medical need for such disease or condition.” (Reuters/Andrew Kelly/File Photo)
Robinette, Navy Wife And as a mother, she was diagnosed with stiff-person syndrome in the spring of 2023.
“This RMAT designation means the science agrees that fast-tracking this treatment can save lives,” she added. “This is very exciting news for the entire autoimmune disease community.”
Click here to get the FOX News app
According to the company, 50 patients suffering from oncology and autoimmune diseases were treated with KYV-101 in clinical trials at more than 15 sites in Europe and the United States.
Kyverna plans to continue collecting data from Phase 2 trials of the drug in stiff-person syndrome, multiple sclerosis, and myasthenia gravis.
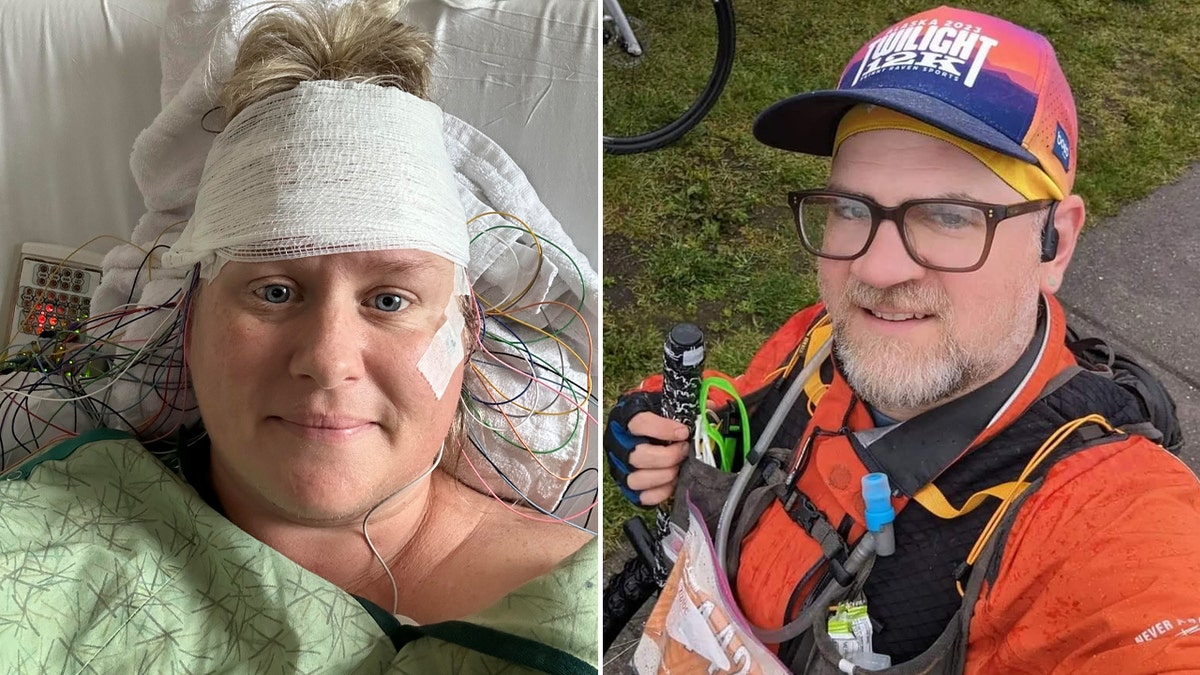
On the left is Carrie Robinette (45) of San Diego, California, and on the right is Corwin Wilkie (44) of Anchorage, Alaska, both of whom have stiff person syndrome. (Carrie Robinette, Corwin Wilkie)
Phase 1 and 2 trials are also underway for systemic sclerosis and lupus.
“we, Sponsored Exams “To advance our knowledge of the potential immunological resetting of patients’ immune systems,” Maag said in a company statement.
For more health related articles, Fox News
Piqué added, “Stiff-person syndrome has devastating and life-altering effects on patients suffering from this rare autoimmune disease…We look forward to seeing data emerging from the KYSA-8 trial, as this trial has the potential to dramatically change the treatment landscape for SPS.”
Fox News Digital has reached out to the FDA for additional comment.

















