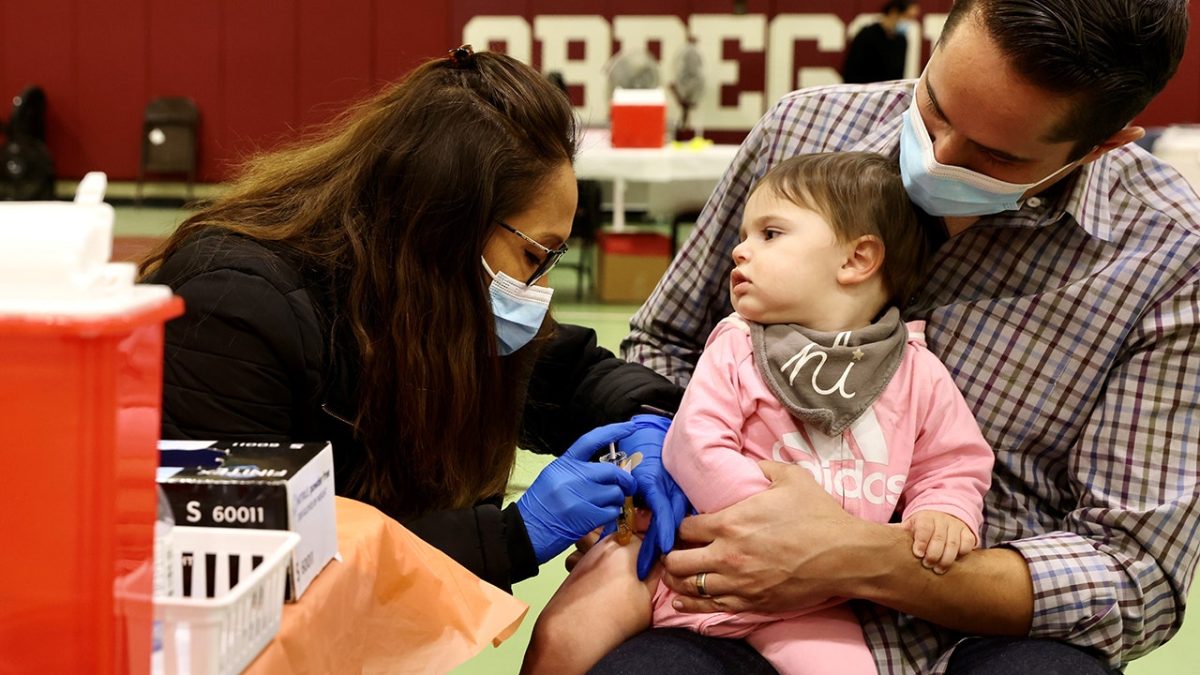
FDA’s Decision on Pfizer Vaccine for Young Children
The U.S. Food and Drug Administration (FDA) has stated that it cannot extend emergency use authorizations for the Pfizer BioNTech COVID-19 vaccine intended for children under five. This development may remove the only COVID-19 vaccine currently accessible for this age group.
A Pfizer representative indicated, “We are exploring possible pathways with our partners and advocating for the EUA for this age group to remain in effect for the 2025-2026 season.” They emphasized that these discussions don’t reflect the safety or effectiveness of vaccines, which still show a favorable profile.
As of now, the Centers for Disease Control and Prevention (CDC) guidelines suggest potential vaccine options for moderately or severely immunocompromised children aged between 6 months and 4 years. Other recommendations will still apply to those aged 5-11 and 12-17 years.
“The COVID-19 pandemic effectively concluded with the end of the federal public health emergency in May 2023,” an HHS spokesperson remarked, mentioning that the department is commenting on future regulatory changes, adding, “Any talk of upcoming actions should be viewed as speculation unless officially announced by HHS.”
Recently, Robert F. Kennedy Jr., director of Health and Welfare (HHS), noted concerns about vaccine availability for healthy children and pregnant women, calling for a transparent process to restore public trust around vaccination discussions.
George Benjamin, executive director of the American Public Health Association, commented on the issue, noting the low vaccination rates but highlighting the potential impacts of the decision. He stated, “It would significantly affect vaccine availability, and I believe making this choice now is untimely.”