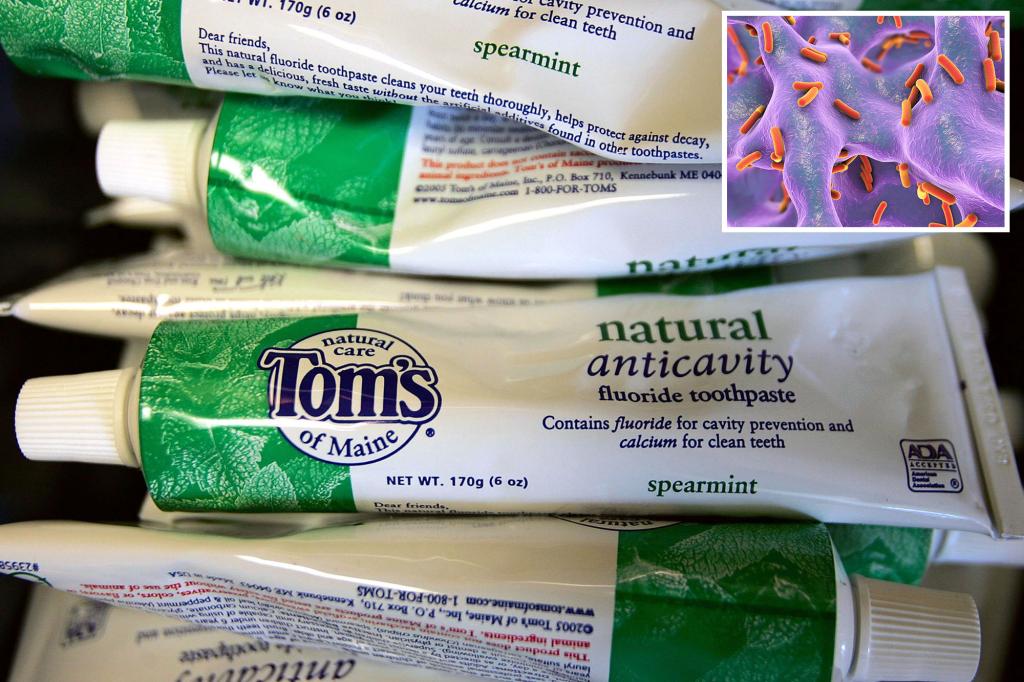
Colgate-Palmolive is facing federal charges for using water contaminated with dangerous bacteria to manufacture the popular Tom's of Maine toothpaste and to clean equipment used to mass produce the product at a major facility. We received a warning from regulators.
The Food and Drug Administration wrote a letter Tuesday to the company, saying it had not done enough to address the problem, saying it first became aware of the problem three years ago at Kennebunk-based Personal Care, which was acquired by Colgate-Palmolive. The company was Tom's of Maine. In 2006.
According to the FDASeveral strains of Pseudomonas aeruginosa were detected in water used to make Tom's of Maine Simply White Clean Mint Paste at the company's Sanford, Maine manufacturing plant between June 2021 and October 2022. was discovered.
“None of these incidents were investigated to evaluate the impact on the product or the performance of the water system,” the FDA wrote.
Pseudomonas aeruginosa, or Pseudomonas aeruginosa, is an antibiotic-resistant pathogen that can cause pneumonia, urinary tract infections, and eye and blood infections.
The agency also wrote that Tom's of Maine reported “numerous multiple incidents” in which the bacterium Ralstonia insidiosa was detected in water used in the manufacturing process.
R. insidiosa can cause respiratory tract and bloodstream infections when exposed to humans.
The FDA said the company's response was “insufficient” and required it to provide “a comprehensive assessment of the design and controls of your manufacturing operations.”
“Water is a key ingredient in many OTC (over-the-counter) medications,” the FDA wrote in its warning letter.
“It is important to have a robustly designed water system and to effectively control, maintain and monitor the system to consistently produce water suitable for pharmaceutical use.”
A spokesperson for Tom's of Maine told the Post that the company is “cooperating with the FDA” and “resolving the issues raised” in the letter.
“We always test finished products before they leave our control, and we continue to have full confidence in the safety and quality of the toothpaste we manufacture,” a company representative told the Post. spoke.
Tom's of Maine says it has “hired water experts to evaluate our systems” at the Sanford plant and has “also implemented additional safeguards to ensure compliance with FDA standards,” There were no problems in the inspection.”
The company also said it is making “capital investments as part of ongoing and significant upgrades to the Sanford plant's water system.”
“Tom's remains committed to creating safe and effective natural products for consumers and maintaining trust in our brand,” a company representative said.
Tom's of Maine has earned a reputation for selling products made with natural ingredients and free of artificial flavors and preservatives.