Expanded Cervical Cancer Screening Guidelines
New federal guidelines are broadening the options available for cervical cancer screenings, moving beyond the often-unpopular Pap smear.
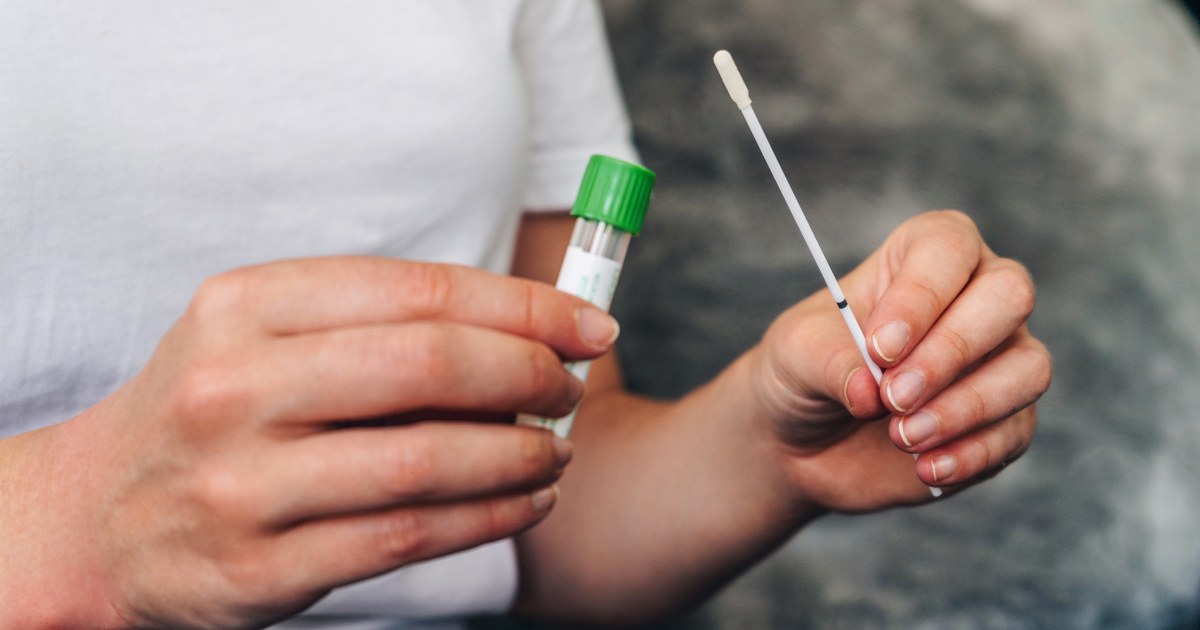
On Monday, the Health Resources and Services Administration, part of the Health and Human Services Department, introduced new recommendations that permit women aged 30 to 65, who are at average risk for cervical cancer, to choose a self-administered HPV test.
This test identifies human papillomavirus, the main cause of cervical cancer—an ailment that results in over 4,000 deaths annually in the U.S.
According to these updated guidelines, women are advised to undergo an HPV test every five years, either through a self-swab or a test performed by a healthcare professional. Although Pap smears should still be offered, they are no longer considered the primary screening method for women over 30. Pap smears are still suggested every three years for those aged 21 to 29.
As most private health insurers are obligated to cover preventive services recommended by HRSA, these insurers are expected to follow the new guidelines by January 1, 2027, including coverage for any necessary follow-up testing.
This new guidance reflects recent research indicating that HPV tests can, in fact, enhance the detection of abnormal cervical cells that may lead to cancer when compared to Pap smears. Previous federal guidelines suggested HPV tests and Pap smears every five years for women aged 30 to 65, or Pap smears every three years.
Using a self-administered HPV test involves inserting a plastic tube—akin to a tampon—into the vagina, followed by twisting the handle to collect cells for lab examination. In contrast, Pap smears utilize a speculum for the clinician to gather cervical cells.
Research has shown that self-swab HPV tests provide similar accuracy to those conducted by a clinician. The Food and Drug Administration has approved two self-swab tests for use in doctors’ offices since 2024, along with an at-home option called the Teal Wand that can be requested after a telehealth visit. This test is priced around $250 out of pocket, although some insurance plans may cover it.
In December, the American Cancer Society updated its recommendations to support self-swab HPV tests every three years for women aged 25 to 65, although it still prefers clinician-administered tests every five years.
While cervical cancer is largely preventable, rates among women in their 30s and early 40s have reportedly been increasing year after year. This rise may stem from missed screenings or women not receiving HPV vaccinations, which have been available since 2006. These vaccines can prevent over 90% of cervical cancers, and regular screenings play a crucial role in identifying HPV infections or precancerous cells.
In an editorial published Monday in the Journal of the American Medical Association, federal health officials indicated that these new guidelines could enhance screening rates, especially for women in rural areas or those with limited access to healthcare facilities. They also noted that some may find self-swab tests more comfortable or preferable for privacy reasons.
It’s worth mentioning that a quarter of women aged 21 to 65 in the U.S. are not current with their cervical cancer screenings, and the rate of screenings in this demographic declined from 47% in 2019 to 41% in 2023 due to the pandemic.
“By reducing testing barriers, expanding choices, empowering women, and eliminating patient cost-sharing, these guidelines for cervical cancer screening represent a significant advancement for women’s health across the United States,” said the health officials.