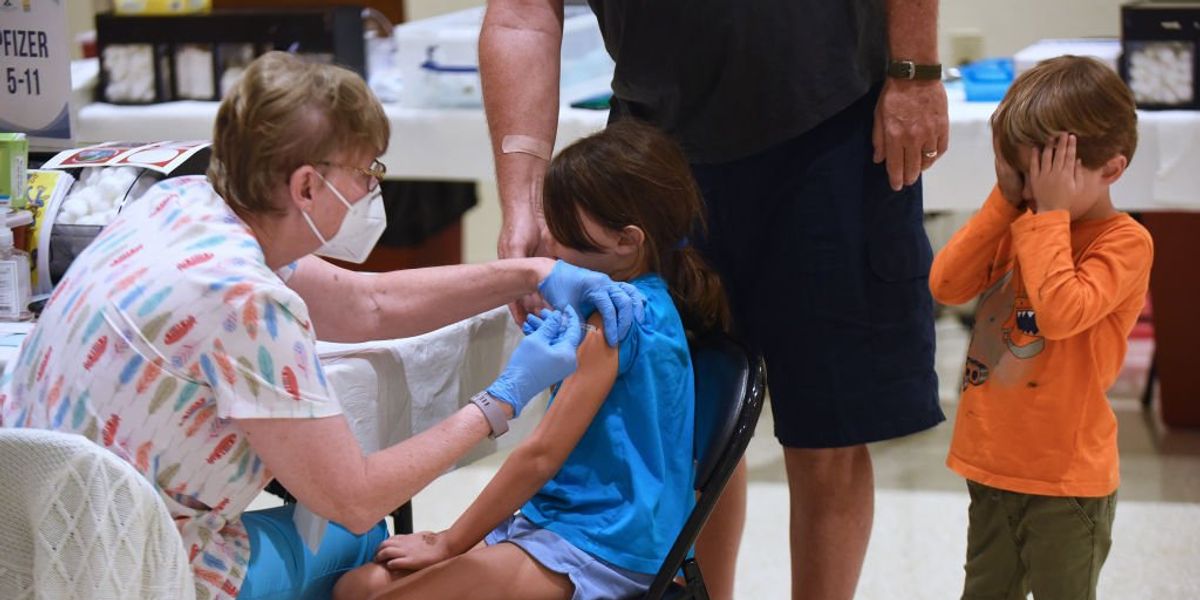
COVID-19 vaccines are completely safe and people are skeptical of claims that they are safe.
effective Claims long made by people who were once trusted Government agency, Pharmaceutical companyand the media In the midst of a historic censorship campaign targeting opponents and critics, their suspicions appear to have been tested once again.
A new peer-reviewed, multinational study examining data from nearly 100 million people finds not only a well-documented link between COVID-19 vaccines and an increased risk of heart disease, but also It also highlighted troubling links between Zeneca, Moderna and Pfizer. vaccines and medical conditions such as Guillain-Barre syndrome, inflammation of the brain and spinal cord, Bell’s palsy, and convulsions.
The study was conducted by the Global COVID-19 Vaccine Safety Project, a global vaccine data network initiative supported by both the Centers for Disease Control and Prevention and the Department of Health and Human Services.
published Last week, it was published in Vaccine Magazine, the official journal of the Japanese Vaccine Society.
Lead author Krystyna Faxova from the Danish National Serum Institute’s Epidemiological Research Department, along with researchers from other countries including Argentina, Australia, Canada, Finland, New Zealand and Scotland, investigated whether neurological diseases are at greater risk. was evaluated. , blood- and heart-related medical conditions that occur after receiving the COVID-19 vaccine.
The actual-versus-expected study, which included data on 99 million people vaccinated against SARS-CoV-2 in eight countries, found that “the risk up to 42 days after vaccination is the background risk for most outcomes. were generally similar, however, several potential safety signals were identified.
“In Bell’s palsy, the OE ratio increased after the first dose. [Pfizer’s] BNT162b2 and [Moderna’s] “mRNA-1273,” the study states, “OE ratio of febrile convulsions after the first and second doses of mRNA-1273 and generalized seizures after the first and fourth doses of BNT162b2 of mRNA-1273.” has also increased.”
Researchers have found that Guillain-Barre syndrome and cerebral venous sinus thrombosis (intracerebral venous sinus thrombosis) can occur after vaccination with viral vector vaccines, such as the Oxford-AstraZeneca COVID-19 vaccine administered in Canada and Europe. We observed a tendency for a type of blood clot (a type of blood clot) to occur.nation
got thrown away Spring 2021 over reports of dangerous blood clots.
Specifically, the researchers found that “there was a statistically significant increase in GBS cases within 42 days after the first ChAdOx1 administration.” They expected him to find 76 her GBS “events”, but he found far more than twice that number.
The University of Auckland, which hosts the Global Vaccine Data Network,
I got it. The researchers said, “Safety signals for transverse myelitis (inflammation of parts of the spinal cord) after viral vector vaccines and acute disseminated encephalomyelitis (inflammation and swelling of the brain and spinal cord) after viral vector and mRNA vaccines.” He said he discovered the possibility of
However, the paper notes, “While some case reports suggest a possible association between COVID-19 vaccination and ADEM, there is no consistent pattern regarding vaccination or post-vaccination timing, and large-scale No potential association has been confirmed in extensive epidemiological studies.”
However, research confirmed
Previously established safety signals For myocarditis and pericarditis after mRNA vaccination, see “Myocarditis after the 1st, 2nd, and 3rd doses of BNT162b2 and mRNA-1273, and the 1st and 4th doses of mRNA-1273; We highlight that the risk of pericarditis after the third dose of ChAdOx1 is significantly higher and within a risk period of 0 to 42 days. ”
The study said that for both viral vector and mRNA vaccines, “the importance of potential safety signals may be underestimated due to potential underreporting across countries.”
“The population size of this study increased our chances of identifying rare potential vaccine safety signals,” Faxoff said.
Said In a statement. “A single site or region is unlikely to have a large enough population to detect very rare signals.”
The researchers argued that “the safety signals identified in this study should be evaluated in terms of their rarity, severity, and clinical relevance.”
Additionally, the research team concluded that the “overall risk” and “Assessing benefits must take into account the risks associated with infectious diseases.” It is twice as infectious as vaccination. ”
Blaze News reported earlier this month that a peer-reviewed study published January 24 in the Springer Nature Group journal Cureus found that COVID-19 vaccines have an “unacceptable harm-to-reward ratio.” It was reported that it was suggested that the product was rushed.
The study found that the types of serious adverse events that would be considered in risk-benefit comparisons were “not caused by COVID-19 mRNA vaccination; “It is often mistakenly thought to be caused by (COVID-19).”
“The misattribution of SAEs to COVID-19 is likely due to the amplification of the negative effects of continued SARS-CoV-2 subvariant infection after mRNA injection,” the study said. “The damage caused by the mRNA product is redundant on both sides.” [post-acute COVID-19 syndrome] Severe acute COVID-19 infections often obscure the pathogenic contribution of vaccines. ”
Do you like Blaze News? Avoid censorship and sign up for our newsletter to get articles like this delivered straight to your inbox. Please register here!