Changes in Covid-19 Vaccine Recommendations for Pregnant Women and Children
The Ministry of Health and Human Services is reportedly set to revise its guidance regarding the Covid-19 vaccine for pregnant women and children. Sources close to the situation suggest that an announcement may come soon, coinciding with the CDC’s efforts to implement a new vaccine approval framework.
While the relevant agencies have not commented on these developments, Dr. Marty McCurry, the US Food and Drug Commissioner, implied earlier this week that significant changes might be forthcoming.
According to McCurry, “You’d want to see evidence that there’s a need for sending another Covid-19 shot to new and healthy children.” He expressed skepticism about the CDC’s push for vaccination among young, healthy kids and mentioned anticipating news on this topic in the near future.
Reportedly, this announcement could come within days.
The CDC has previously recommended that anyone who hasn’t been vaccinated in the last six months should consider receiving the Covid-19 vaccine.
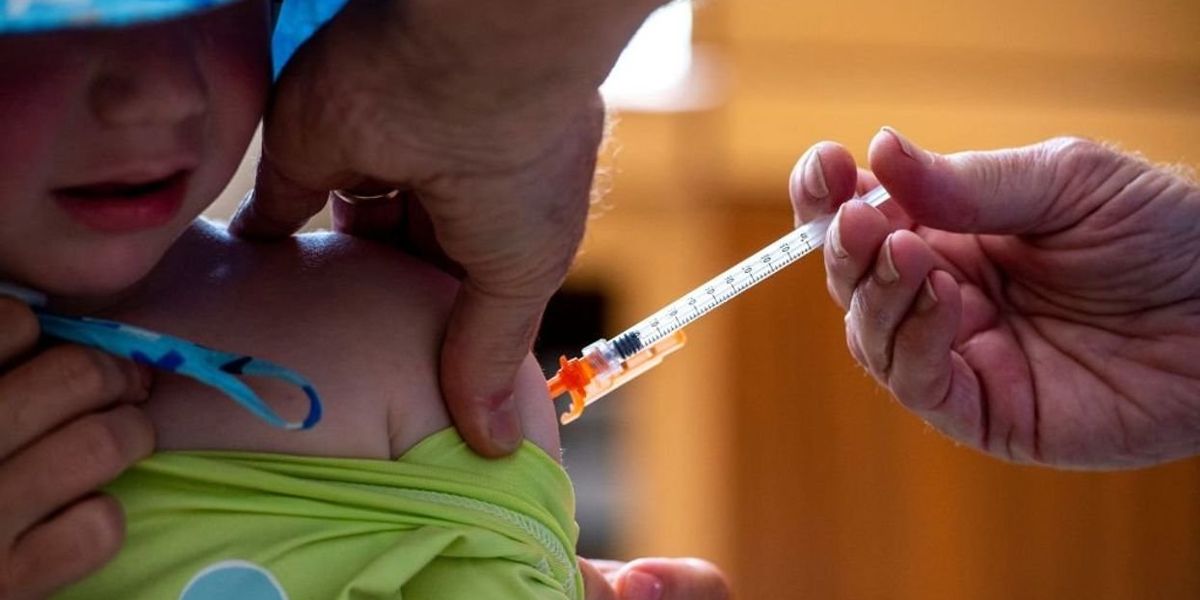
The agency’s messaging highlights the importance of vaccinations for the elderly, pregnant women, and those planning to conceive, particularly urging parents to vaccinate children between six months and four years old with two or three doses of the Pfizer vaccine.
As of April 26, approximately 14.4% of pregnant women had received the 2024-25 Covid-19 vaccine, with just 13% of children aged six months to 17 years similarly vaccinated, according to CDC data.
Despite the CDC’s steadfastness in previous recommendations, some authorities, including Florida’s surgeon general, have raised concerns regarding the vaccine’s safety and effectiveness. Research suggests that the vaccine may not be as safe as originally portrayed, particularly for kids and teens, who might be able to go without the vaccine’s risks.
Last month, a study from the Florida Department of Health indicated that adults who received the Pfizer vaccine could face a heightened risk of various health issues compared to those vaccinated with Moderna.
Additionally, a report from the Global Covid Vaccine Safety Project highlighted troubling associations between vaccines from AstraZeneca, Moderna, and Pfizer and various medical conditions.
Another peer-reviewed study linked Covid-19 vaccinations to severe myocarditis, particularly in younger populations. It suggested that current risks associated with the vaccines might outweigh their benefits.
Dr. Peter McCullough, a noted vaccine critic, commented that nearly five years post the vaccine rollout, there might be promising news for women and children regarding the postponement of these recommendations.
Former HHS Secretary Robert F. Kennedy Jr. previously called for revoking approval for the Covid-19 vaccine entirely, particularly emphasizing the need to protect children and pregnant women.
Unfortunately, Kennedy’s warnings did not lead to immediate changes. In the closing weeks of the Biden administration, the HHS extended liability protections for Covid-19 vaccine manufacturers until the end of 2029.
Recent actions taken by HHS reflect movements initiated during the Trump presidency, including alterations in vaccine recommendations.
As the vaccine landscape continues to shift, experts like Paul Offit believe we could witness a gradual dismantling of the current vaccine infrastructure. Others, like epidemiologist Tara Smith, suggest that changes in recommendations could impact insurance coverage for vaccines.