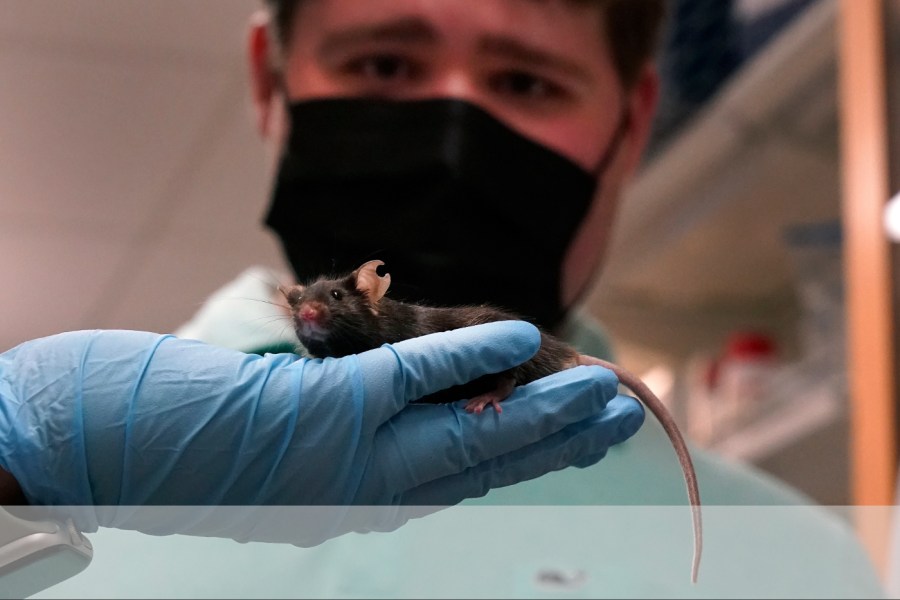
The Food and Drug Administration and the National Institutes of Health held a joint workshop on July 7. The surprising takeaway was the announcement that “NIH no longer seeks proposals specifically for animal models,” which indicates a significant shift in their approach to research.
This move aligns with the FDA’s plan to phase out animal testing requirements for drugs. There’s a notable transition happening, where the focus is shifting away from animal research towards “more effective and human-related methods.”
Essentially, this means that the FDA might allow the introduction of new drugs to people without the usual prior animal testing, while the NIH intends to limit funding for types of research that have historically led to significant medical breakthroughs.
From the perspective of a biomedical researcher who utilizes mouse models, this raises concerns. Eliminating animal testing could jeopardize drug safety and set back years of crucial biomedical advancements.
If we think about drug regulations, our ability to identify new treatments really hinges on insights gained from preclinical animal studies. These studies are foundational for understanding drug relevance, safety, and potential effectiveness before they reach human trials.
Skipping this crucial step would make the journey of discovering new drugs far more complex, and, frankly, their success rates would likely decline. Typically, after initial studies, drugs enter clinical testing, which involves a progressively larger human cohort across three phases before approval.
Animal models play a crucial role in biomedical research. They help us explore unpredictable biological effects across different organ systems. For instance, a cancer treatment might kill tumor cells in a dish but may also cause unforeseen heart issues or trigger harmful immune responses in a living organism. Testing on animals is the closest we can get to assessing a drug’s safety and efficacy before it hits human bodies. If developers are allowed to bypass this, the first living subjects for new drugs would, unfortunately, become humans.
Moreover, researchers rely on animals to decode diseases and create entirely new treatments. Mice, often referred to as the “immunological Rosetta Stone,” have informed much of our current understanding of the immune system.
Consider advancements like cancer immunotherapy or treatments for autoimmune disorders and allergies. These wouldn’t exist without years of foundational research conducted with animal models.
About half of NIH-funded grants involve some form of animal use. So, what prompts the world’s largest public funder of biomedical research to instigate such a radical change? The reasoning is rooted in the belief that new human-based methods—such as laboratory-grown human models and computational tools—might serve as better alternatives. While these can certainly complement existing animal models, treating them as viable substitutes might be premature.
A common critique of animal research is that results don’t always translate directly to humans, which is true. However, there’s scant evidence to support that the suggested alternatives would provide more reliable outcomes.
Instead of completely abandoning animal research, we should consider refining current models to more accurately reflect human biology.
It’s acknowledged that some animal models need updates. For example, a drug for immune cells that passed animal trials back in 2006 led to crises for several healthy volunteers in human clinical trials.
Later investigations revealed that the laboratory mice used had weaker immune systems due to their sterile environment, which actually hampered their immune development. Interestingly, introducing a common pet store mouse into the lab mice’s environment helped better mimic the human immune system.
In subsequent tests of that same drug with the improved mice, researchers noticed similar inflammatory complications as observed in human trials. Innovative approaches using these enhanced mice are already contributing to a better understanding of the side effects of cancer treatments.
This advancement has inspired researchers to improve mouse models, potentially accelerating biomedical discoveries.
After the announcement, animal rights advocates declared it a “groundbreaking move,” representing a crucial step towards modernizing scientific research and reducing animal suffering.
However, ethical treatment of animals doesn’t have to imply complete exclusion from scientific processes. The NIH has held to strict guidelines since the early 1900s, ensuring proposals include considerations for alternatives, justifications for animal use, and measures to minimize distress.
Animal testing remains essential for discovering new treatments. Consider the story of KJ, a newborn diagnosed with a severe genetic disorder that heavily impacts survival rates. Custom treatments involving modified mouse models played a key role in developing a suitable therapy within eight months.
There’s a long history of biomedical successes tied to animal models—think about the foundations for understanding brain and heart functions or the breakthroughs in insulin and cancer therapies.
Human health should always be a priority. It’s crucial for scientists, medical professionals, policymakers, and the public to work together to uphold the research systems that have enabled monumental health advancements.
As Nobel Prize winner Aldem Pataputian recently asserted, “Protecting American science means ensuring shared prosperity for the future.”
The FDA and NIH are open to public feedback on these developments and future workshops, and it’s important that people share their opinions.