Data Reporting
Statistical methods for determining sample size were not applied prior to the experiments. The study lacked randomization, and the investigators did not maintain blinding during both experimental procedures and the assessment of outcomes.
Materials
The substances used included alvimopan, naloxone, mitragynine pseudoindoxyl, DAMGO, and loperamide, all sourced from Cayman Chemicals and MedChemExpress. Detergents were obtained from Anatrace. The radioligand [3H]naltrexone, with a specific activity of 48.19 Ci per mmol, was provided by the National Institute on Drug Abuse Drug Supply Program. Other general laboratory reagents and chemicals were purchased from Millipore Sigma unless indicated otherwise.
Constructs
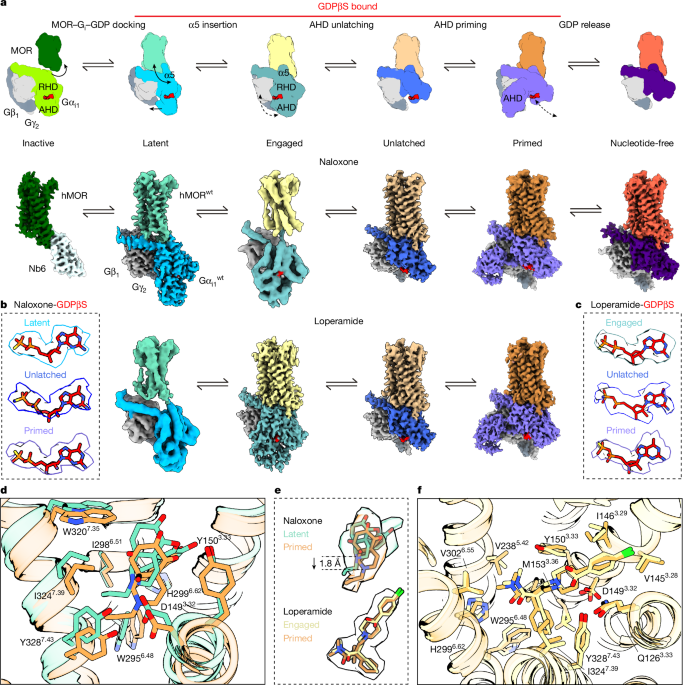
For the expression of recombinant proteins, full-length human MOR was subcloned into an adapted pFastBac1 vector featuring an N-terminal Flag, a 10xHis tag, and thermostabilized b562RIL along with a C-terminal TwinStrep tag and another 10xHis tag. Both terminal tags have HRV-3C protease cleavage sites to allow for their removal. In the case of the inactive MOR structure, two mutations (M264L and K2696.24R) were introduced to facilitate Nb6 binding.
Transient transfections for all BRET assays of the wild-type full-length human MOR used the pcDNA 3.1(−) vector in HEK293F cells, under the control of the cytomegalovirus promoter. These receptor constructs included an N-terminal haemagglutinin signal peptide and a Flag tag. For TRUPATH and GloSensor assays, the MOR construct was untagged. In the RG-BRET assay, Rluc8 was added immediately following the C terminus of the wild-type MOR construct.
During TRUPATH assays, a tricistronic vector encoding Gαi1, Gβ1, and Gγ2 was employed, where GFP2 was linked to the N terminus of Gγ2 and Rluc8 was introduced after residue G90 of the Gαi1 subunit. RG-BRET assays utilized a Gαi1β1γ2 construct, with GFP2 at the N terminus of Gγ2. The control plasmid pOZITX-S1 was provided by J. Javitch.
Cell Culture and Transfections
HEK293F cells were grown in FreeStyle293 Expression Medium at 37 °C with 5% CO2, shaking at 110 rpm. For transfections, cells were seeded a day in advance and transfected at a density of 1.0 × 106 cells per ml using polyethyleneimine with a DNA:PEI ratio of 1:2. For RG-BRET assays, MOR–Rluc8 and Gαi/Gβ1/Gγ2–GFP2 constructs were transfected in a 1:2 ratio. In TRUPATH BRET assays, the ratios were adjusted accordingly.
BRET2 Assays
Assays with live cells involved washing cells with an assay buffer and transferring them to white-bottom 96-well plates. For permeabilization assays, collected cells were washed with KPS permeabilization buffer and treated with digitonin and apyrase as needed, then transferred into 96-well plates containing desired ligands in ligand buffer.
Measurements were taken using a PHERAstar FSX multimode plate reader, with specific filters for BRET2. Coelenterazine 400a was added just prior to measuring with Rluc8. NetBRET signals were calculated based on emission intensities at given wavelengths without further normalization.
Every assay was run at least three separate times, with three data points per experiment. EC50 and IC50 values were derived from raw results using GraphPad Prism.
cAMP Inhibition Assays
After 48 h post-transfection, cells were washed and placed into 96-well plates. Ligands were prepared as solutions in assay buffer, and following an incubation period, luciferin was added to measure cAMP production triggered by β2 adrenergic activation.
Radioligand Saturation Binding Assays
These assays utilized membrane fractions from HEK293F cells expressing human MOR. Membranes were prepared by resuspending cell pellets in a hypotonic buffer and centrifuged to yield membrane fractions. The concentration of membrane proteins was determined, then stored for later use.
Competition assays were conducted in 96-well plates, where membrane fractions were combined with naltrexone and a range of ‘cold’ doses, then processed through vacuum filtration and counted using a plate reader.
MOR Expression and Purification
The recombinant expression of MOR constructs was executed in Sf9 insect cells using the Bac-to-Bac system. Cell pellets post-expression were resuspended, treated with buffers, and subjected to ultracentrifugation for membrane isolation.
For purification, membranes were lysed, and the debris removed, followed by affinity purification with anti-Flag resin. Final solutions were concentrated and later used for forming complexes with other proteins.
Expression and Purification of Nb6M
The Nb6 plasmid was transformed into E. coli and grown under specific conditions until expression was induced. Following centrifugation, the cells were lysed and the NB6 protein purified using affinity columns.
Expression and Purification of NabFab
C43 cells were similarly transformed and induced to express NabFab. Cell lysates underwent clarifying treatments before being applied to purification columns.
Expression and Purification of G Proteins and scFv16
G-proteins were co-expressed in insect cells, collected, and purified based on established procedures. The scFv16 was produced and purified using similar methods, involving several purification steps.
Complexation of MOR and G Protein Heterotrimers
Purified MOR was mixed with Gαiβγ heterotrimer complexes, followed by processing for a clear structure which then underwent further purification to prepare for cryoEM studies.
CryoEM Sample Preparation, Data Collection and 3D Reconstruction
Samples for cryoEM were prepared and flash-frozen for imaging. Data was collected using sophisticated imaging techniques and analyzed for subsequent reconstruction efforts.
Single Particle CryoEM Image Processing
Data processing was carried out utilizing advanced software, with rigorous steps to ensure quality and resolution in the resultant images.
Model Building and Refinement
Models were constructed using existing crystal structures as references, ensuring precise fitting into density maps, followed by thorough validation of model geometry.
MD Simulations
MD simulations were managed with GROMACS, leveraging specific force-field parameters. The simulations assessed the dynamics of protein-lipid interactions among other factors.
Data Statistical Analysis
Statistical analysis of the data was performed using standard techniques with significance levels appropriately set.
Figures and Graphical Illustrations
Various graphical designs and illustrations were created using reliable software tools, ensuring clarity in data representation.
Reporting Summary
Further details regarding the research design can be found in the accompanying Supplementary Material.