Advancements in Spinal Cord Injury Treatment
Experimental drugs are showing promise in aiding movement for individuals suffering from spinal cord injuries. One such drug, NVG-291—an injectable peptide—has undergone a phase 2 trial with participants reporting significant improvements.
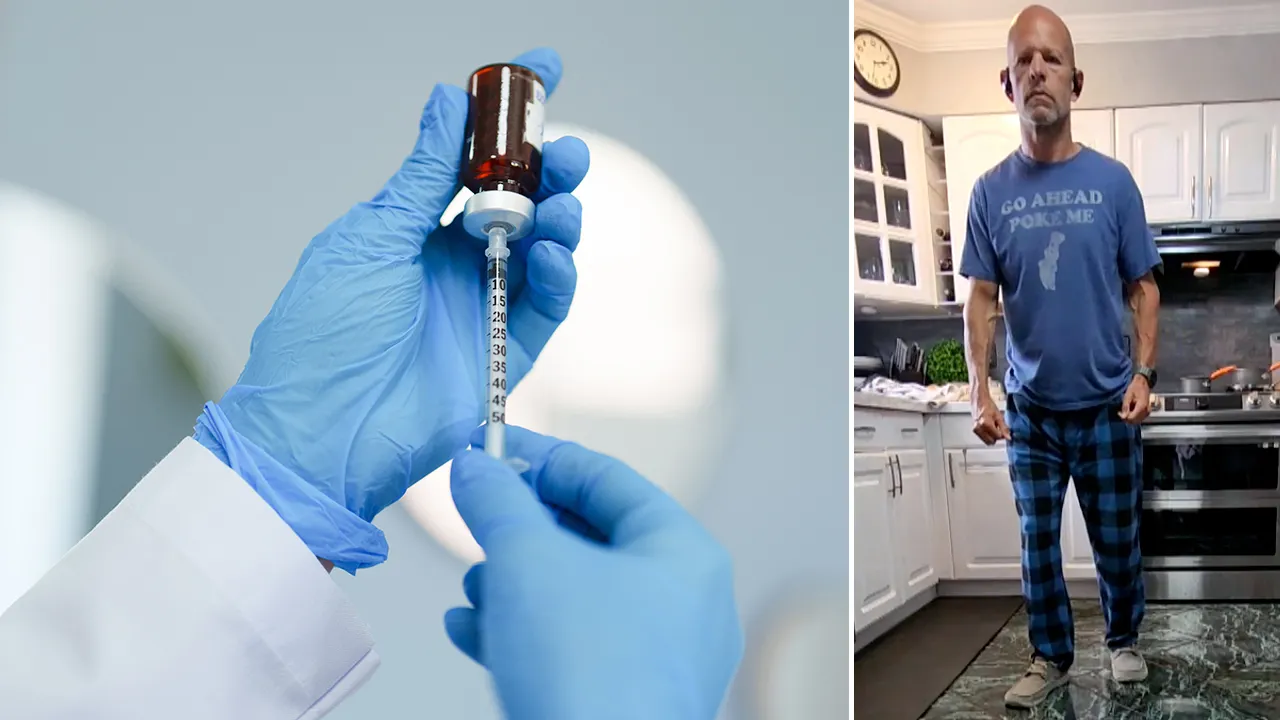
Larry Williams, a participant from Philadelphia, shared his experience of regaining the ability to walk following a severe accident that led to his paralysis. At 58, he had a harrowing encounter with a tree while riding on a small mountain road. Although he wore a helmet, he sustained serious injuries, breaking vertebrae in his neck.
After undergoing spinal surgery, Williams faced two weeks of paralysis. Fortunately, after beginning treatment, he noticed the movement slowly returning.
Initially, he could only walk with assistance, struggling with mobility, particularly with his hands, and he even lost about 40 pounds. After researching potential treatments, he found the NVG-291 trial and was accepted as a participant.
From April 2024, he received daily injections of the drug for three months, along with an hour of physical therapy focused on hand exercises and treadmill walking.
At the conclusion of the trial, Williams was able to walk 10 meters in a time of fifteen seconds, showing remarkable progress. Even though he hasn’t taken the drug since July 2024, he is still experiencing ongoing improvements over a year later.
“I’m not really putting in a lot of effort. I’m not currently in treatment,” Williams mentioned. “Just a few days ago, I managed to get up and balance for around thirty seconds.” He reflected on how just six months prior, he couldn’t lift his feet off the ground for three seconds.
Since participating in the trial, he has noted advancements in his walking abilities, and he has even resumed swimming. “I’m reaching out to others with similar injuries. It’s like many have gotten stuck where I was, and I feel fortunate for this opportunity. I hope to regain independence in my life,” he expressed.
Williams remarked that performing physical tasks has become “easier and quicker,” highlighting improvements in the smoothness and unrestricted movement of his legs.
Understanding NVG-291
Dr. Monica Perez, a scientific expert at Shirley Ryan AbilityLab and professor at Northwestern University, elaborated on the drug’s mechanism. Initially tested in animals, NVG-291 was shown to enhance motor functions. The subsequent human clinical trials randomized participants, with half receiving the drug designed to improve nervous system function.
This peptide is significant as it helps in removing obstructions preventing nerve growth after injury. Essentially, following a spinal injury, the body typically communicates signals that halt nerve fiber growth. NVG-291 interferes with these signals, paving the way for potential regeneration.
While traditional approaches to spinal cord injuries often involve therapies like stem cells, this newer peptide method is considered more straightforward, can be administered at home, and appears to be quite safe according to Perez.
Research continues to explore the long-term effects of NVG-291, as noted by Perez. Although they observed substantial changes in nerve functionality compared to a placebo group, the longevity of the drug’s effects remains uncertain.
As there are currently no FDA-approved treatments for spinal cord injuries, the commitment to advancing this research is strong. Williams encourages others with similar conditions to consider this treatment: “It could make a real difference for individuals like me who are facing injury challenges. I truly hope others get this chance to reclaim their lives.”